Class 12 Chemistry NCERT Chapter 2: Higher Order Thinking Skills (HOTS) Questions
Below are some Important Questions on Solutions that will help students tackle complex problems with ease. To understand the concepts better, students can also refer to Class 12 Solutions notes.
Question 1: $\mathrm{HA}(\mathrm{aq}) \rightleftharpoons \mathrm{H}^{+}(\mathrm{aq})+\mathrm{A}^{-}(\mathrm{aq})$
The freezing point depression of a 0.1 m aqueous solution of a monobasic weak acid HA is $0.20^{\circ} \mathrm{C}$. The dissociation constant for the acid is
Given :
$\mathrm{K}_{\mathrm{f}}\left(\mathrm{H}_2 \mathrm{O}\right)=1.8 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$, molality $\equiv$ molarity
(1) $1.38 \times 10^{-3}$
(2) $1.1 \times 10^{-2}$
(3) $1.90 \times 10^{-3}$
(4) $1.89 \times 10^{-1}$
Answer:
$\begin{aligned} & \Delta \mathrm{T}_{\mathrm{f}}=\mathrm{i} \mathrm{K}_{\mathrm{f}} \mathrm{m} \\ & \mathrm{i}=\frac{\Delta \mathrm{T}_f}{\mathrm{~K}_{\mathrm{f}} \cdot \mathrm{m}} \\ & \mathrm{i}=\frac{0.20}{1.8 \times 0.1}=1.11 \\ & \mathrm{i}=1.11 \\ & \alpha=\frac{i-1}{\mathrm{n}-1}(\text { for } \mathrm{HA}, \mathrm{n}=2) \\ & \alpha=\frac{1.11-1}{1}=0.11 \\ & \mathrm{~K}_{\mathrm{a}}=\frac{c \alpha^2}{1-\alpha}=\frac{0.1 \times(0.11)^2}{1-0.11}=1.38 \times 10^{-3}\end{aligned}$
Hence, the correct answer is option (1).
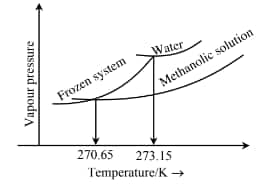
Question 2: When ' $x$ ' $\times 10^{-2} \mathrm{~mL}$ methanol (molar mass $=32 \mathrm{~g}$; density $=0.792 \mathrm{~g} / \mathrm{cm}^3$ ) is added to $100 \mathrm{~mL}$ water (density $=1 \mathrm{~g} / \mathrm{cm}^3$ ), the following diagram is obtained.
![Depression in freezing point]()
$x=\ldots \ldots . . . . . . . . .$. (nearest integer)
[Given: Molal freezing point depression constant of water at $273.15 \mathrm{~K}^2$ is $1.86 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ]
i) 543
ii) 559
iii)550
iv)540
Answer:
$\begin{aligned} & \Delta \mathrm{T}_{\mathrm{f}}=273.15-270.65=2.5 \mathrm{~K} \\ & \Delta \mathrm{T}_{\mathrm{f}}=\mathrm{K}_{\mathrm{f}} \mathrm{m} \Rightarrow 2.5=1.86 \times \frac{\mathrm{n}}{0.1} \\ & \Rightarrow \mathrm{n}=0.1344 \text { moles } \\ & \Rightarrow \mathrm{w}=0.1344 \times 32=4.3 \mathrm{~g} \\ & \text { Volume }=\frac{4.3}{0.792}=5.43 \mathrm{ml}=543 \times 10^{-2} \mathrm{ml}\end{aligned}$
Hence, the correct answer is option (1).
Question 3: Given below are two statements :
Statement (I) : Molal depression constant $\mathrm{K}_{\mathrm{f}}$ is given by $\frac{M_1 R T_f}{\Delta S_{f u s}}$, where symbols have their usual meaning.
Statement (II) : $\mathrm{K}_{\mathrm{f}}$ for benzene is less than the $\mathrm{K}_f$ for water.
In the light of the above statements, choose the most appropriate answer from the options given below :
(1) Statement I is incorrect but Statement II is correct
(2) Both Statement I and Statement II are incorrect
(3) Both Statement I and Statement II are correct
(4) Statement I is correct but Statement II is incorrect
Answer:
Statement-I
Molar depression constant $\mathrm{k}_f=\frac{\mathrm{M}_{\mathrm{i}} \mathrm{RT}_{\mathrm{f}}^2}{\Delta \mathrm{H}_{\text {fius }}}$
$\begin{aligned}
& \mathrm{k}_f=\frac{\mathrm{M}_1 \mathrm{RT}_{\mathrm{f}}}{\left[\frac{\Delta \mathrm{H}_{\text {fis }}}{\mathrm{T}_{\mathrm{f}}}\right]} \\
& \mathrm{k}_f=\frac{\mathrm{M}_1 \mathrm{RT}_f}{\Delta \mathrm{~S}_{\text {fus }}}
\end{aligned}$
Hence statement-I is correct
but $\mathrm{k}_{\mathrm{f}}$ for benzene $=5.12 \frac{{ }^{\circ} \mathrm{C}}{\text { molal }}$
$\mathrm{k}_{\mathrm{f}}$ for water $=1.86 \frac{{ }^{\circ} \mathrm{C}}{\text { molal }}$ Hence statement- II is incorrect
Hence, the correct answer is option (4).
Question 4: The vapour pressure of an aqueous solution is found to be 750 torr at a temperature $T$, and the same solution shows an elevation in boiling point equal to 1.04 K . If $T$ is the boiling point of pure water, then the atmospheric pressure should be ( $K_b$ of water $=0.52 \mathrm{Kkg} / \mathrm{mol}$ )
Answer:
$\Delta \mathrm{T}_{\mathrm{b}}=\mathrm{K}_{\mathrm{b}} \mathrm{~m} \Rightarrow \mathrm{~m}=1.04 \mathrm{~K} / 0.52 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}=2 \mathrm{~mol} \mathrm{~kg}^{-1}$
For $1 \mathrm{~kg} \mathrm{H}_2 \mathrm{O}$ :
$\mathrm{n}_{\text {solvent }}=1000 \mathrm{~g} / 18 \mathrm{~g} \mathrm{~mol}^{-1}=55.56 \mathrm{~mol}$
$\mathrm{n}_{\text {solute }}=2 \mathrm{~mol}$
Mole-fraction of solvent
$X_w=\frac{55.56}{55.56+2}=0.965$
Raoult: $P_{\text {solution }}=X_w P^{\circ}$
Given $P_{\text {solution }}=750$ torr,
$P^{\circ}=\frac{750}{0.965} \approx 7.77 \times 10^2 \text { torr. }$
Atmospheric pressure $=P^{\circ} \approx 777$ torr.
Hence, the answer is 777 torr.
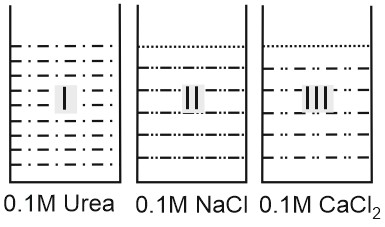
Question 5: Choose the correct observation
![]()
(1) Vapour pressure of solution $I$ is lowest
(2) Relative lowering of vapour pressure is maximum in III
(3) Freezing point is maximum for III
(4) Boiling point is minimum for II
Answer:
$\begin{array}{ccc}
\text { (I) } & \text { (II) } & \text { (III) } \\
\text { Urea } & \mathrm{NaCl} & \mathrm{CaCl}_2 \\
c=0.1 M & 0.1 M & 0.1 M \\
i=1 & i=2 & i=3
\end{array}$
1. Vap. pressure $\propto \frac{1}{i}$
$\begin{gathered}
\therefore \quad V \cdot P \quad I>I I>I I I \\
\frac{\Delta p}{p_0} \propto i \\
\text { 2. } \quad \therefore \frac{\Delta p}{p_0} \quad I I I>I I>I \\
\text { 3. } \Delta T_f=i k_f \cdot m . \\
\Delta T_f \propto i \\
T_f^0-T_f \propto i \\
\therefore I I I<I I<I
\end{gathered}$
4.$\begin{aligned}
& \Delta T_b=i K_b \cdot m \\
\end{aligned}$
$T_b-T_b^0 \propto i \quad \therefore I I I>I I>I$
Hence, the correct answer is option (2).
Approaches to Solve Questions of Chapter 2
To solve Solutions question effectively, students should focus on understanding the core concepts, formulas, and their applications. NCERT Exemplar Class 12 Solutions Chapter 2 questions will strengthen problem-solving skills.
1. The solve questions of this chapter first step is to identify the type of solution, whether the solution is solid in liquid, liquid in liquid or gas in liquid.
2. The next step is to understand the basic terms frequently used in almost every question, like solute, solvent and concentration.
3. Concentration terms like molarity, molality, mass per cent, mole per cent, parts per million are often asked in exams, and the questions are interrelated with these concentration terms. So, proper knowledge of these concentration terms is a must.
4. Then we must have the knowledge of topics like:
- Solubility
- Ideal solution
- Factors Affecting Solubility like the identification of the effect of Temperature and Pressure.
University of York, Mumbai
UG & PG Admissions open for CS/AI/Business/Economics & other programmes.
University of Bristol, Mumbai Enterprise Campus
Bristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes
5. Students must be aware of the Colligative properties, like:
- Relative lowering of vapour pressure
- Elevation of Boiling Point
- Depression of Freezing point
6. A Proper understanding of basic concepts and practice helps students clear their doubts and solve questions effectively for NCERT Exemplar Class 12 Chemistry Solutions Chapter 2 Solutions. While finalising answer, do not forget to verify whether units are correct and also check for significant figures.
Formulas for Class 12 Chemistry Chapter 2
Some important formulas of chapter 2 questions are given below.
1. Mass percentage (w/w)
Mass percent $=\frac{\text { Mass of the component in the solution }}{\text { Total mass of the solution }} \times 100$
2. Volume percentage (V/V):
Volume $\%$ of a component $=\frac{\text { Volume of the component }}{\text { Total volume of solution }} \times 100$
3. Mass by volume percentage (w/V):
Mass by Volume $\%(\mathrm{w} / \mathrm{V})=\left(\frac{\text { Mass of solute }(\mathrm{g})}{\text { Volume of solution }(\mathrm{mL})}\right) \times 100$
4. Parts per million:
$\begin{aligned} & \text { Parts per million }= \\ & \frac{\text { Number of parts of the component }}{\text { Total number of parts of all components of the solution }} \times 10^6\end{aligned}$
5. Mole fraction:
Mole Fraction =$\frac{\text { Number of moles of the component }}{\text { Total number of moles of all the components }}$
For example, in a binary mixture, if the number of moles of $A$ and $B$ are $n_{\mathrm{A}}$ and $n_{\mathrm{B}}$ respectively, the mole fraction of A will be
$x_{\mathrm{A}}=\frac{n_{\mathrm{A}}}{n_{\mathrm{A}}+n_{\mathrm{B}}}$
For a solution containing i number of components, we have:
$
x_i=\frac{n_i}{n_1+n_2+\ldots \ldots+n_i}=\frac{n_i}{\sum n_i}
$
6. Molarity:
Molarity $=\frac{\text { Moles of solute }}{\text { Volume of solution in litre }}$
7. Molality:
Molality $(\mathrm{m})=\frac{\text { Moles of solute }}{\text { Mass of solvent in } \mathrm{kg}}$
8. Raoult's law
$P_{\text {solution }}=($ Mole fraction of solvent $) \times($ Vapour pressure of pure solvent $)$
9. Dalton's law of partial pressures
$P_{\text {total }}=P_1+P_2+P_3+\ldots$
10. Relative lowering of vapour pressure :
By Raoult's law,
$\frac{P^0-P}{P^0}=X_{\text {solute }}$
11. Elevation of boiling point :
$\Delta T_b=K_b \cdot m$
where $m$ is molality of solution and $K_b$ is called boiling point elevation
12. Depression of freezing point :
$\Delta T_f=K_f \cdot m$
$K_f=$ Molal depression constant
13. Osmosis and osmotic pressure :
$\Pi=C R T$
14. van't Hoff factor $(i)$
$i=\frac{\text { Observed colligative property }}{\text { Calculated colligative property }}$
15. Relation between van't Hoff factor and degree of dissociation
$\alpha=\frac{i-1}{n-1}$
16. Relation between van't Hoff factor and degree of association
$\alpha=\frac{1-i}{1-1 / n}$