University of Liverpool, Bengaluru Campus
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Have you ever thought about how light can behave both like a wave and a particle? Or how tiny particles like electrons can show wave-like behavior? That is what Chapter 11: Dual Nature of Radiation and Matter in Class 12 Physics is all about. These ideas are the basics of modern physics. These NCERT exemplar solutions are prepared by expert faculty as per the latest syllabus
The NCERT Exemplar Solutions for Class 12 Physics Chapter 11 by Careers360 offer step-by-step answers to all exemplar problems. These include multiple-choice questions(MCQs), short answers and long form problems all aligned with the latest CBSE syllabus. Solving these NCERT exemplar for class 12 physics questions will help you understand important ideas better, boost your analytical thinking and prepare effectively for exams like CBSE Boards, JEE, and NEET.
This Story also Contains

Question:1
Answer:
The answer is the option (d)
Question:2
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly
(a) 1.2 nm
(b) 1.2 x 10-3 nm
(c) 1.2 x 10-6 nm
(d). 1.2 x 10 nm
Answer:
The answer is the option (b)Question:3
Consider a beam of electrons (each electron with energy E0) incident on a metal surface kept in an evacuated chamber. Then
(a) no electrons will be emitted as only photons can emit electrons
(b) electrons can be emitted but all with an energy, E0
(c) electrons can be emitted with any energy, with a maximum of E0 – $\phi$ ($\phi$ is the work function)
(d) electrons can be emitted with any energy, with a maximum of E0
Answer:
The answer is the option (d) When a beam of electrons (each electron with energy E0 ) incident on a metal surface is kept in an evacuated chamber, energy of incident electrons will be transferred to the emitted electrons due to elastic collisions. A part of E0 of incident electrons is consumed against work function to emit the electrons. Thus, the maximum energy of emitted electrons is E0Question:4
Consider Fig. 11.7 in the NCERT textbook of physics for Class XII. Suppose the voltage applied to A is increased. The diffracted beam will have the maximum at a value of θ that
(a) will be larger than the earlier value
(b) will be the same as the earlier value
(c) will be less than the earlier value
(d) will depend on the target
Answer:
The answer is the option (c)Question:5
A proton, a neutron, an electron and an α-particle have the same energy. Then their de Broglie wavelengths compare as
$(a) \lambda _{p} = \lambda _{n} > \lambda _{e} > \lambda _{\alpha} $
$ (b) \lambda _{\alpha} < \lambda _{p} = \lambda _{n} < \lambda _{e}$
$ (c) \lambda _{e} < \lambda _{p} = \lambda _{n} > \lambda _{\alpha} $
$ (d) \lambda _{e} = \lambda _{p} = \lambda _{n} = \lambda _{\alpha}$
Answer:
The answer is the option (b)Question:6
An electron is moving with an initial velocity v = v0i and is in a magnetic field B = B0j. Then, its de-Broglie wavelength
(a) remains constant
(b) increases with time
(c) decreases with time
(d) increases and decreases periodically
Answer:
The answer is the option (a)Question:7
An electron (mass m) with an initial velocity v = v0i(v0 > 0) is in an electric field $E=E_{0}\hat{i}$ (E0 = constant > 0). Its de-Broglie wavelength at time t is given by
$(a) \frac{\lambda_{0}}{\left [ 1+\frac{eE_{0}}{m}\frac{t}{v_{0}} \right ]}$
$ (b)\lambda_{0}\left [ 1+\frac{eE_{0}t}{mv_{0}} \right ]$
$(c)\lambda_{0}$
$ (d)\lambda_{0}t$
Answer:
The answer is the option (a)Question:8
An electron (mass m) with an initial velocity $v=v_{0}\hat{i}$ is in an electric field $E=E_{0}\hat{i}$ IF $\lambda_{0}=\frac{h}{mv_{0}}$ its de-broglie wavelength at time t is given by
$(a)\lambda_{0}$
$(b)\lambda_{0}\sqrt{1+\frac{e^{2}E_{0}^{2}t^{0}}{m^{2}v_{0}^{2}}}$
$(c)\frac{\lambda_{0}}{\sqrt{1+\frac{e^{2}E_{0}^{2}t^{0}}{m^{2}v_{0}^{2}}}}$
$(d)\frac{\lambda_{0}}{\left (1+\frac{e^{2}E_{0}^{2}t^{0}}{m^{2}v_{0}^{2}} \right )}$
Answer:
The correct answer is option (c)Question:9
Relativistic corrections become necessary when the expression for the kinetic energy 1/2 mv2 , becomes comparable with mc2, where m is the mass of the particle. At what de Broglie wavelength will relativistic corrections become important for an electron?
$(a)\lambda=10nm$
$ (b)\lambda=10^{-1}$
$ (c)\lambda=10^{-4}$
$ (d)\lambda=10^{-6}\\$
Answer:
The answer is the option (c, d)Question:10
Two particles $A_{1}$ and $A_{2}$ of masses $m_{1}$, $m_{2}$ ($m_{1}$ >$ m_{2}$) have the same de Broglie wavelength. Then
(a) their momenta are the same
(b) their energies are the same
(c) energy of $A_1$ is less than the energy of $A_2$
(d) energy of $A_1$ is more than the energy of $A_2$
Answer:
The answer is the option (a. c)Question:11
The de Broglie wavelength of a photon is twice the de Broglie wavelength of an electron. The speed of the electron is ve = c/100. Then
(a) Ee/Ep = 10-4
(b) Ee/Ep = 10-2
(c) Pe/mec = 10-2
(d) Pe/mec = 10-4
Answer:
The answer is the option (b,c)$\begin{gathered}
\lambda=\frac{h}{p}=\frac{h}{\sqrt{2 m E}}=\frac{h}{\sqrt{2 m q V}} \\
\lambda_{\text {Neutron }}=\frac{0.286 * 10^{-10}}{\sqrt{E}} m=\frac{0.286 * 10^{-10}}{\sqrt{E}} A^{\circ}
\end{gathered}$
At ordinary temperature, the energy of thermal neutrons
$\begin{aligned}
& \quad E=k T \Rightarrow \lambda=\frac{h}{\sqrt{2 m k T}} \\
& \lambda_{\text {Thermal } \text { Neutron }}=\frac{30.83}{\sqrt{T}} A^{\circ} \\
& \text { Mass of electron }=m_e \\
& \text { Mass of photon }=m_p
\end{aligned}$
de Broglie wavelength of the electron
$\begin{aligned}
& \quad \lambda_e=\frac{h}{m_e v_e}=\frac{100 h}{m_e c} \\
& K E_e=\frac{1}{2} m_e v_e^2 \\
& m_e v_e^2=\sqrt{2 E_e m_e} \\
& \lambda_e=\frac{h}{m_e v_e}=\frac{h}{\sqrt{2 E_e m_e}} \\
& E_e=\frac{h^2}{2 \lambda_e^2 v_e}
\end{aligned}$
de Broglie wavelength of the proton is $\lambda_p$
$\begin{aligned}
& E_p=\frac{h c}{\lambda_p}=\frac{h c}{2 \lambda_e} \\
& \frac{E_p}{E_e}=\left(\frac{h c}{\lambda_e}\right) *\left(\frac{2 \lambda_e^2 m_e}{h}\right)=100 \\
& p_e=m_e v_e=m_e * \frac{c}{100} \\
& \frac{p_e}{m_e c}=10^{-2}
\end{aligned}$
Question:12
Photons absorbed in the matter are converted to heat. A source emitting n photon/sec of frequency ν is used to convert 1kg of ice at 0°C to water at 0°C. Then, the time T taken for the conversion
(a) decreases with increasing n, with ν fixed
(b) decreases with n fixed, ν increasing
(c) remains constant with n and ν changing such that nν = constant
(d) increases when the product nν increases
Answer:
The answer is the option (a, b. c).Question:13
A particle moves in a closed orbit around the origin, due to a force which is directed towards the origin. The de-BrOglie wavelength of the particle varies cyclically between two values $\lambda_{1},\lambda_{2}$ with $\lambda_{1}>\lambda_{2}$ Which of the following statements are true?
(a) The particle could be moving in a circular orbit with origin as centre
(b) The particle could be moving in an elliptic orbit with origin as its focus
(c) When the de-Broglie wavetength is $\lambda_{1}$ the particle is nearer the origin than when its value is $\lambda_{2}$
(d) When the de-Broglie wavelength is $\lambda_{2}$ the particle is nearer the origin than when its value is $\lambda_{1}$
Answer:
The answer is the option (b,d)Get your results instantly with our calculator!
Question:14
Answer:
It is given that the proton and $\alpha$-particle are accelerated at theQuestion:15
(i) In the explanation of photoeletric effect, we assume one photon of frequency v collides with an electron and transfers its energy. This leads . to the equation for the maximum energy Emax of the emitted electron as $E_{max} = hv - \phi_{0}$
where $\phi_{0}$ is the work function of the metal. If an electron absorbs 2 photons (each of frequency v), what will be the maximum energy for the emitted electron?
(ii) Why is this fact (two photon absorption) not taken into consideration in our discussion of the stopping potential?
Answer:
(i) In the question, 2 photons transfer its energy to one electron as E=hvQuestion:16
Answer:
With an increase in the wavelength of the photon, there is a decrease in the frequency or increase in the energy of the photon. Let us consider 2 possible cases:Question:17
Do all the electrons that absorb a photon comes out as photo electrons?
Answer:
A common observation in the photoelectric effect is that most of the electrons knocked by photons are scattered into the metal by absorbing photons. In order to escape from the surface, the electron must absorb enough energy to overcome the positive ion attraction in the material of the surface.Question:18
Answer:
Let EX and EV be the energies given by one photon in X-rays andQuestion:19
Answer:
There is a transfer of momentum to the atoms on the metal surface when the photons strike the surface. This also results in a decrease in speed of the photons. Thus, the momentum of photons is transferred to the nucleus and electrons of the metal.Question:20
Answer:
Let the maximum energies of emitted electrons be K1 and K2 when 600 nm and 400 nm visible light are used according to questionQuestion:21
Answer:
As the electrons rotate in a circular path,Question:22
Answer:
$\begin{aligned} & I_A=I_B(\text { Given in the question }) \\ & n_A h v_A=n_B h v_B \\ & n_A=2 n_B[\text { Given }] \\ & 2 n_B v_A=n_B v_B \\ & 2 v_A=v_B\end{aligned}$Question:23
Answer:
The de-Broglie wavelength is given byQuestion:24
Answer:
According to the Bragg’s law of diffraction, condition for the nth maxima is given byQuestion:25
Answer:
Area (A)=10-2 cm2=10-4m2Question:26
Answer:
Question:27
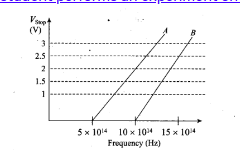
(i) Which material A or B has a higher work function?
(ii)Given the electric charge of an electron = 1.6 x 10-19C, find the value of h obtained from the experiment for both A and B. Comment on whether it is consistent with Einstein's theory.
Answer:
We know that $\phi_{0}=hv_{0}$ where v0=Threshold frequencyQuestion:28
Answer:
When the collision is elastic, law of conservation of momentum is followed.Question:29
Consider a 20 W bulb emitting light of wavelength 5000 $A^{\circ}$ and shining on a metal surface kept at a distance 2m. Assume that the metal surface has work function of 2 eV and that each atom on the metal surface can be treated as a circular disk of radius 1.5 $A^{\circ}$ .
(i) Estimate no. of photons emitted by the bulb per second. [Assume no other losses]
(ii) Will there be photoelectric emission?
(iii) How much time would be required by the atomic disk to receive energy equal to work function (2 eV)?
(iv) How many photons would atomic disk receive within time duration calculated in (iii) above?
(v) Can you explain how photoelectric effect was observed instantaneously?
Answer:
Given that $\mathrm{P}=20 \mathrm{~W}$
$\begin{aligned}
& \lambda=5000 A^{\circ}=5000 * 10^{-10} \mathrm{~m} \\
& d=2 \mathrm{~m} \\
& \phi=2 \mathrm{ev} \\
& r=1.5 * 10^{10} \mathrm{~m}
\end{aligned}$
i) Number of photon emitted by bulb per second= $\frac{p \lambda}{h c}=5 * 10^{19} \mathrm{sec}$
ii Energy of the incident photon= $\frac{h c}{\lambda}=2.48 \mathrm{eV}$
iii Time required by the atomic disk to receive energy is=28.4 sec
Let the time spent be $\Delta T$
$E=P * A * \Delta t=P * \pi r^2 \Delta t$
Energy transferred by the bulb in full solid angle to atoms= $ 4 \pi d^2 \phi$
$\begin{aligned}
& p * \pi r^2 \Delta t=4 \pi d^2 \phi \\
&\Delta t=\frac{4 d^2 \phi}{P r^2}=\frac{4 * 2 * 2 * 2 * 1.6 * 10^{-10}}{20 * 1.5 * 1.5 * 10^{-10} * 10^{-10}} \sec =\frac{12.8 * 10^{-19+20}}{5 * 2.25}
\end{aligned}$
(iv) Number of photons received by the atomic disk $=\mathrm{N}$
$\begin{aligned}
\frac{n_1 \pi r^2 \Delta t}{4 \pi d^2}&=\frac{n_1 r^2 \Delta t}{4 d^2} \\
&= \frac{5 * 10^{19} * 1.5 * 1.5 * 10^{-20} * 11.4}{4 * 2 * 2} \\
&\approx 0.80 \cong 1 \text { photon per atom }
\end{aligned}$
(iv) We say that photoelectric emission is instantaneous as it involves a collision between the incident photon and free electron lasting for a very short span of time, say $\leq 10^{-9} \mathrm{sec}$
|
Chapter 11 Dual Nature of Radiation and Matter |
Frequently Asked Questions (FAQs)
These questions are solved keeping in mind the CBSE pattern and how the marks are divided as per the steps.
Yes, one can download these questions so that one uses the solutions offline.
Try to understand the NCERT exemplar solutions for Class 12 Physics chapter 11 and connect them to the topics that one has studied in the chapter.
One will learn in detail about wave theory of light, particle nature of light, photoelectric effect, Davisson and Germer experiment, etc.
On Question asked by student community
Hello Shubham,
NCERT Class 12 Chemistry chapter-wise notes and solutions are available at the link given below. Keep checking for updated uploads when NCERT PYQs (Previous Year Questions) are published.
https://school.careers360.com/ncert/ncert-solutions-class-12-chemistry
Hi!
Given below is the link to access CBSE Class 12 Sociology Question Paper 2026 with Answer Key:
https://school.careers360.com/boards/cbse/cbse-class-12-sociology-answer-key-2026
Hello Hashi,
Download the CBSE Class 12 Business Studies 2026 paper from the link below to prepare effectively.
https://school.careers360.com/boards/cbse/cbse-class-12-business-studies-question-paper-2026
Hello Gilla
You can download the question paper from the link given below:
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers-class-10-maths
Hope it helps.
Hello Student,
Check the article given below to access and download the CBSE question paper for classes 10 and 12.
Link:
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Apply for UG & PG programmes from Victoria University, Delhi NCR Campus
Admissions open for UG & PG programs at Illinois Tech Mumbai
Apply for UG & PG courses at University of Aberdeen, Mumbai Campus
UG & PG Admissions open for CS/AI/Business/Economics & other programmes.
Bristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes
