Aakash Repeater Courses
Take Aakash iACST and get instant scholarship on coaching programs.
Ever wondered what happens when ammonia meets alkyl or aryl groups? Let us find out the answer through amines! Amines are a class of organic compounds which is derived by replacing one or more hydrogen atoms of ammonia with alkyl or aryl groups. The chapter will make students learn about different types, structures, properties and reactions of amines. The topics like Hofmann bromamide degradation, Gabriel phthalimide synthesis and how amines react in various situations are well discussed in this chapter.

The chapter is important for anyone looking to do well in organic chemistry. The NCERT solutions for class 12 chemistry chapter 9 offer clear explanations that will help students understand these topics and improve their problem-solving skills. These NCERT solutions will help you prepare for boards as well as for competitive exams. We have also included higher-order thinking skills (HOTS) questions to check your critical thinking and deepen your understanding of the topics.
Also Read
Students can download the NCERT Solutions for Chapter 9 in PDF format for free. These solutions are designed to help you understand the fundamental concepts and solve textbook questions with ease.
The detailed solutions to in-text questions are given below
Page No. 262
Question 9.1 Classify the following amines as primary, secondary or tertiary:
Answer :
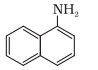
The nitrogen atom is directly connected with only one carbon atom, so it is a primary aromatic amine.
Question 9.1 Classify the following amines as primary, secondary or tertiary:
Answer :
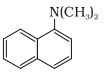
In this compound, the nitrogen atom is directly connected to 3 carbon atoms. So, it is a tertiary aromatic amine
Question 9.1 Classify the following amines as primary, secondary or tertiary:
Answer :
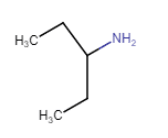
Here, the N atom is directly connected to only one carbon atom. So it is a primary aliphatic amine.
Question 9.1 Classify the following amines as primary, secondary or tertiary:
Answer :
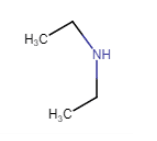
In structure, we can see that N is directly connected to 2 carbon atoms. So, it is a secondary amine.
Question 9.2 (i) Write structures of different isomeric amines corresponding to the molecular formula,
Answer :
different isomeric amines corresponding to the molecular formula,
(i)
(ii)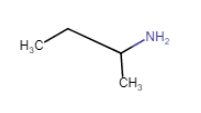 (vi)
(vi) 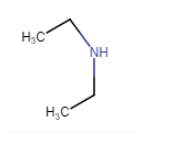
(iii)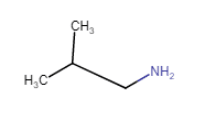 (vii)
(vii) 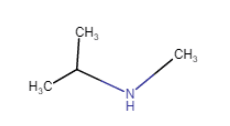
(iv)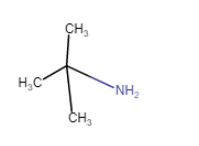 (viii)
(viii) 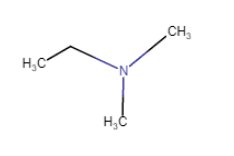
Question 9.2 (ii) Write IUPAC names of all the isomers.
Answer :
IUPAC name of all the isomers-
|
|
Butanamine
|
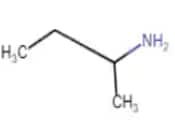 | Butan-2-amine |
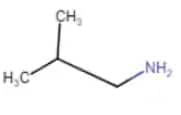 |
2-Methylpropanamine
|
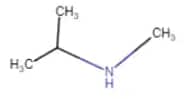 |
N-Methylpropan-2-amine
|
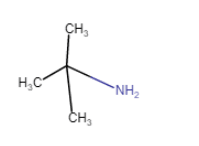 |
2-Methylpropan-2-amine
|
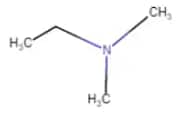 |
N,N-Dimethylethanamine
|
|
N-Methylpropanamine
| |
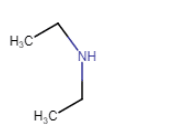 | N-Ethylethanamine |
Question 9.2 (iii) What type of isomerism is exhibited by different pairs of amines?
Answer :
|
|
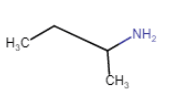
Butanamine
(Chain isomerism + position isomerism) |
 |
Butan-2-amine
(chain isomerism + position isomerism) |
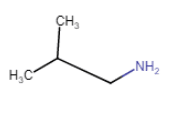 |
2-Methylpropanamine
(chain isomerism) |
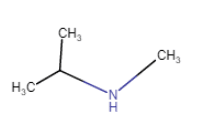 |
N-Methylpropan-2-amine
(position isomerism + metamerism) |
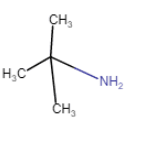 |
2-Methylpropan-2-amine
(chain isomerism) |
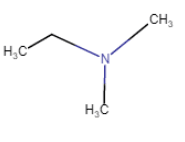 |
N,N-Dimethylethanamine
|
|
|
N-Methylpropanamine
(Position isomerism) |
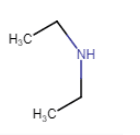 |
N-Ethylethanamine
(no isomerism) |
Question 9.3 (i) How will you convert
Answer :
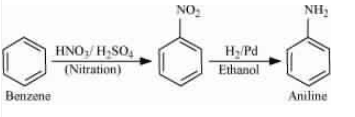
Nitration of benzene gives nitrobenzene. And after that, reduce the nitro group by catalytic hydrogenation.
Question 9.3 (ii) How will you convert
Benzene into N, N-dimethylaniline
Answer :
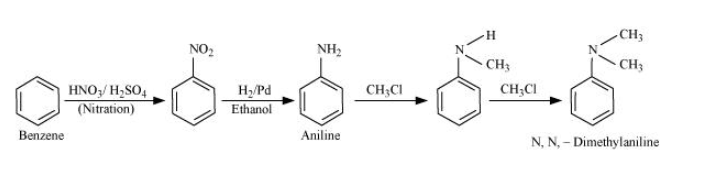
Nitration of benzene gives nitrobenzene, and after catalytic hydrogenation of nitrobenzene, it gives aniline. Aniline reacts with two moles of chloromethane to form N, N-dimethylaniline.
Question 9.3(iii) How will you convert
Answer :
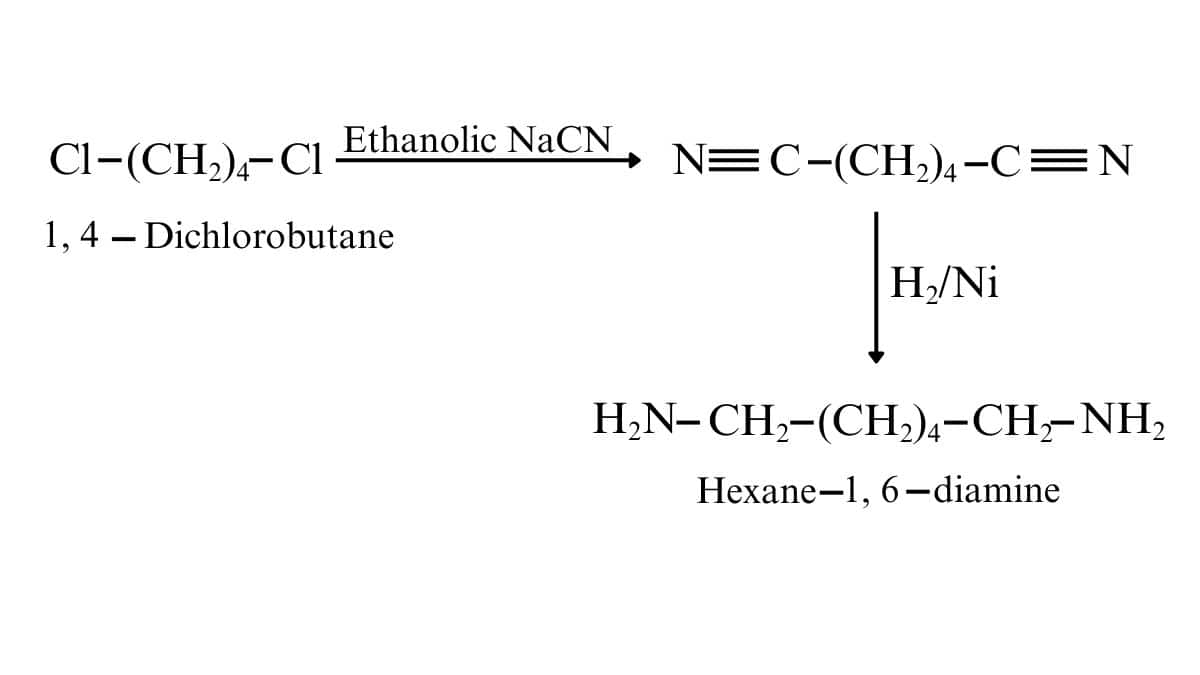
On reacting the given reactant with ethanolic sodium cyanide, the CN molecules replace both chlorine atoms. And after that catalytic hydrogenation, we get our desired product. (reduce the CN to
Question 9.4 (i) Arrange the following in increasing order of their basic strength:
Answer :
Considering the inductive effect, solvation effect and steric hindrance of the alkyl group, which decides the basic strength of alkylamines. The order of basic strength in ethyl-substituted amine is-
and order in the benzene-substituted ring-
Due to the -R effect of benzene
Question 9.4 (ii) Arrange the following in increasing order of their basic strength:
Answer :
On considering the inductive effect, solvation effect and steric hindrance of the alkyl group, the increasing order of basicity in ethyl as a substituted group is shown above.
Question 9.4 (iii) Arrange the following in increasing order of their basic strength:
Answer :
On considering the inductive effect, solvation effect and steric hindrance of the alkyl group, which decides the basic strength of alkyl amines in the aqueous state. The order of basic strength in the case of methyl-substituted amines is -
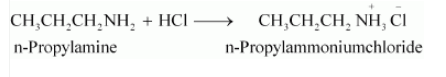
Question 9.5 (i) Complete the following acid-base reactions and name the products:
Answer :
The above-given reaction is an acid-base reaction. So salt is formed.
Question 9.5 (ii) Complete the following acid-base reactions and name the products:
Answer :
It is an acid-base reaction, so the N will accept H from
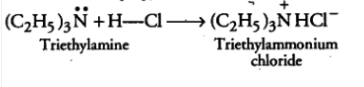
Answer :
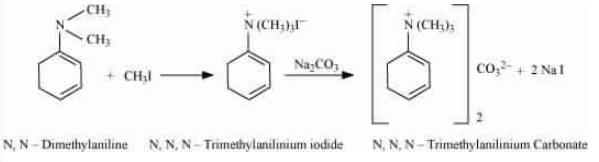
The methyl iodide reacts with aniline to give N, N-dimethylaniline.
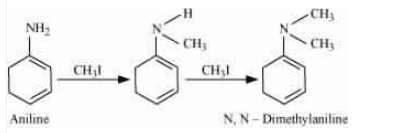
With the excess of methyl iodide in the presence of sodium carbonate solution (
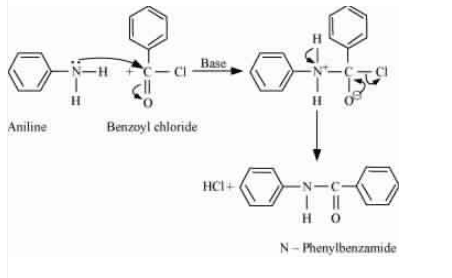
Question 9.7 Write chemical reaction of aniline with benzoyl chloride and write the name of the product obtained.
Answer :
When aniline reacts with benzoyl chloride, HCl is produced as a by-product and N-phenylbenzamide is produced as a major product. The lone pair of the nitrogen atom attacks the acidic carbon of benzoyl chloride.
Answer :
Structures of different isomers corresponding to the molecular formula
|
(i)
|
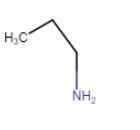 |
Propan-1-amine
|
|
(ii)
|
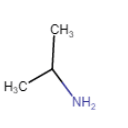 |
Propan-2-amine
|
|
(iii)
|
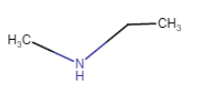 |
N – Methylethanamine
|
|
(iv)
|
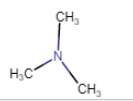 |
N,N-Dimethylmethanamine
|
The structures (i) and (ii) will liberate nitrogen gas (
Question 9.9 (i) Convert
3-Methylaniline into 3-nitrotoluene
Answer :
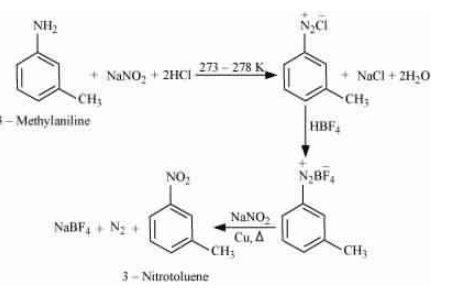
On diazotisation, reaction
Question 9.9 (ii) Convert
Aniline into 1,3,5-tribromobenzene.
Answer :
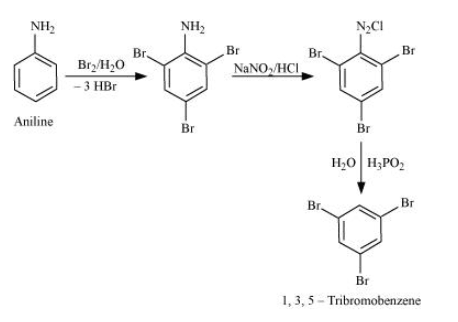
Aniline on bromination in the presence of water gives 2,4,6 tribromoaniline, which on further reacting with nitrous acid converts
The exercise questions are solved here with detailed explanations.
Question 9.1 Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer :
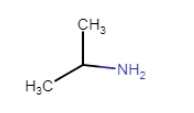
The IUPAC name of the compound is Propan-2-amine
Since
Question 9.1 (ii) Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer :
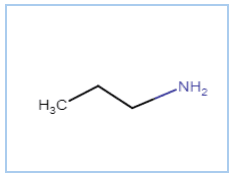
The IUPAC name is Propan-1-amine
It is a primary amine (nitrogen connects with only one carbon atom)
Here is the structure of the compound
Question 9.1 (iii) Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer :
N-Methylpropan-2-amine
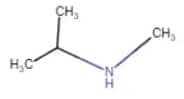
Since N atom is connected with two carbon atoms, it is a two-degree amine.
Question 9.1(iv) Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer :
2-methyl-propan-2-amine
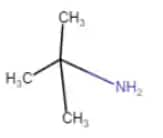
It is a one-degree amine
Question 9.1 (v) Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer :
N-Methylbenzamine or N-methylaniline
Here N atom is connected to two C atoms. So, it is a secondary amine.
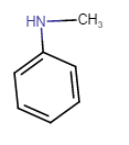
Question 9.1 (vi) Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer:
N,N-Diethylmethylamine
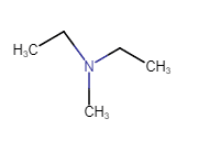
N is connected with three carbon atoms so that it is a tertiary amine
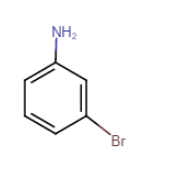
Question 9.1 (vii) Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
Answer :
3-bromoaniline or 3-bromobenzenamine
It is a primary amine
Question 9.2 Give one chemical test to distinguish between the following pairs of compounds.
(i) Methylamine and dimethylamine
Answer :
Carbylamine test
This test is used to distinguish between aliphatic and aromatic primary amines. Heating with chloroform
Here, methylamine is primary and dimethylamine is a secondary amine.
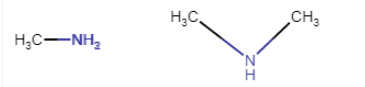
The first one is 1-degree and the second one is 2-degree amine.
Question 9.2 Give one chemical test to distinguish between the following pairs of compounds.
(ii) Secondary and tertiary amines
Answer :
Secondary and tertiary amines can be distinguished by reacting them with Hinsberg's reagent, which is also called benzenesulphonyl chloride.
In the case of a secondary amine
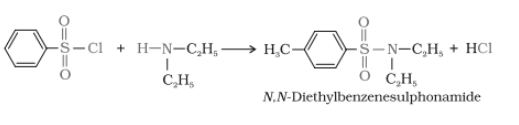
Tertiary amine + benzenesulphonyl chloride
Question 9.2 Give one chemical test to distinguish between the following pairs of compounds.
Answer :
Ethylamine and aniline can be differentiated by the azo-dye test.
On reacting aniline with
Question 9.2 Give one chemical test to distinguish between the following pairs of compounds.
Answer :
Benzylamine reacts with nitrous acid to form a diazonium salt, which is unstable, and it also gives alcohol with the evolution of nitrogen gas. On the other hand, aniline reacts with nitrous acid to form a stable diazonium salt.
Question 9.2 Give one chemical test to distinguish between the following pairs of compounds.
(v) Aniline and N-methylaniline.
Answer :
Aniline and N-methylaniline can both be distinguished by the carbylamine test. Aniline is the primary aromatic compound so it gives s positive test, but N-methyl aniline is secondary and does not give this test.
Structure of both compounds-

Question 9.3 Account for the following:
(i) pKb of aniline is more than that of methylamine.
Answer:
Here is the structure of aniline (left) and methylamine
Due to the resonance in aniline, the electrons of the nitrogen atoms are delocalised over the benzene ring. So, because of that, the electron density at the nitrogen atom decreases and is less available for donation.
On the other hand, in the case of methylamine, the methyl (
Question 9.3 Account for the following:
(ii) Ethylamine is soluble in water whereas aniline is not.
Answer:
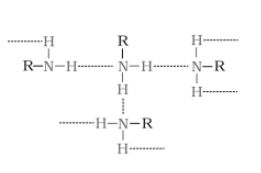
The extent of intermolecular hydrogen bonding in ethylamine is very high. Hence, it is soluble in water. But aniline does not undergo hydrogen bonding with water to a large extent due to the presence of a bulky hydrophobic group
Question 9.3 Account for the following:
(iii) Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
Answer :
Due to the +I effect of the methyl (
Ferric chloride (
The hydroxide ion (
Question 9.3 Account for the following:
Answer :
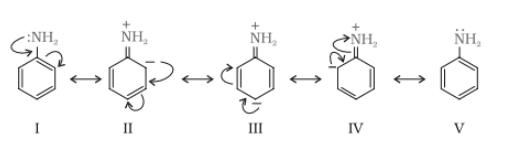
Nitration is carried out in a strongly acidic medium, and aniline is protonated to form the anilinium ion, which is meta-directing. Because of this reason, aniline on nitration gives a substantial amount of meta-nitroaniline.
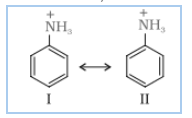
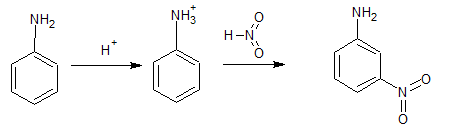
Question 9.3 Account for the following:
(v) Aniline does not undergo Friedel-Crafts reaction.
Answer :
In the Friedel-Crafts reaction, we use
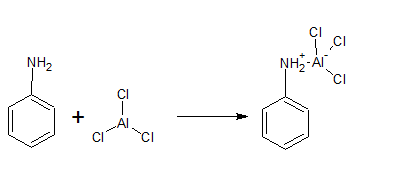
Due to the positive charge on the N-atom. The electrophilic substitution in the benzene ring is deactivated.
Question 9.3 Account for the following:
(vi) Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Answer :
In diazonium salt, the structure undergoes resonance due to which the dispersal of positive charge is more and we know that the higher the resonance, the higher is the stability. Therefore, the diazonium salt of aromatic amines is more stable than that of aliphatic amines.
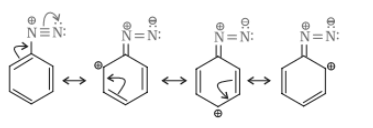
Question 9.3 Account for the following:
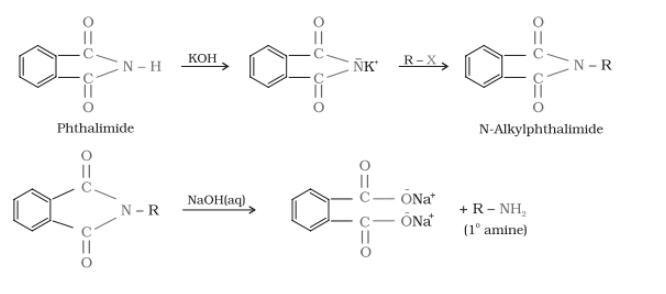
(vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Answer :
Gabriel synthesis is used for primary amines. Secondary and tertiary amines are not formed by this method. Therefore, to obtain pure and only 1-degree amine, the Gabriel phthalimide reaction is preferred.
Question 9.4 Arrange the following:
(i) In decreasing order of the pKb values:
Answer :
Between
Question 9.4 Arrange the following:
(ii) In increasing order of basic strength:
Answer :
The increasing order of basic strength-
Aliphatic amines are more basic than aromatic amines due to the presence of a high electron density of electron at the nitrogen atom so that it can donate an electron. On the other hand, in aromatic amines, the lone pair of electrons is delocalised in the benzene ring, so the availability of electrons is less.
Question 9.4 Arrange the following:
(iii) In increasing order of basic strength:
(a) Aniline, p-nitroaniline and p-toluidine
Answer :
Structure of the following compounds-
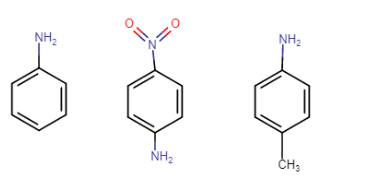
Aniline, p-nitroaniline, p-toluidine
In p-toluidine, the methyl group increases the electron density at the N atom (+I effect of methyl), so it is more basic than aniline. In p-nitroaniline, the nitro group is an electron-withdrawing group, so it decreases the electron density at nitrogen, so it is less basic than aniline.
Question 9.4 Arrange the following:
(iii) In increasing order of basic strength:
Answer :
The increasing order of basic strength-
Question 9.4 Arrange the following:
(iv) In decreasing order of basic strength in gas phase:
Answer :
Decreasing order of basic strength in the gas phase-
In the gas phase, there is no solvation effect. So, the basicity directly depends on the number of electron-donating groups. More is the electron electron-donating groups, the higher the +I effect and if the size of the donating group is large +I effect should also be high.
Question 9.4 Arrange the following:
(v) In increasing order of boiling point:
Answer :
The boiling point of the compounds depends on the extent of hydrogen bonding of the compound. The more the extent, the higher the boiling point.
Thus, the increasing order of boiling point is-
Question 9.4 Arrange the following:
(vi) In increasing order of solubility in water:
Answer :
The more extensive the hydrogen bonding, the greater is the solubility in water.
Thus, the increasing order of solubility in water is-
Question 9.5 How will you convert:
(i) Ethanoic acid into methanamine
Answer :
Ethanoic acid on reacting with
Question 9.5 How will you convert:
(ii) Hexanenitrile into 1-aminopentane
Answer :
First, hexanenitrile undergoes acid hydrolysis, converting the nitrile group (-CN) into a carboxylic acid
Finally, the Hofmann bromamide degradation is carried out, which removes one carbon atom and produces the desired primary amine as the final product.
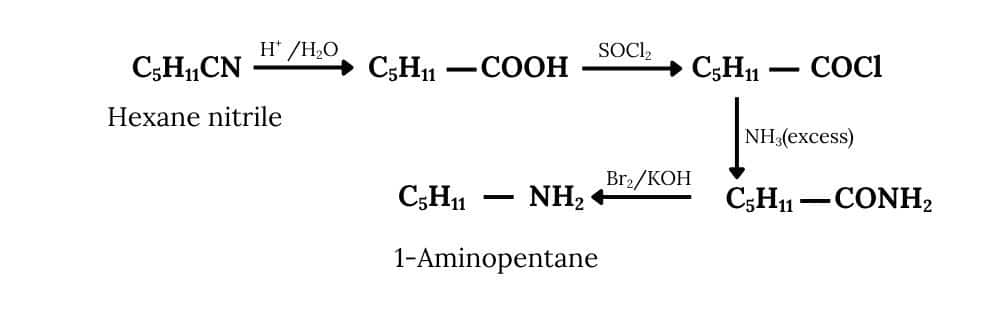
Question 9.5 How will you convert:
(iii) Methanol to ethanoic acid
Answer :
When methanol reacts with phosphorus pentachloride, OH- is reduced by Cl- to form chloromethane, which on reacting with ethanolic sodium cyanide, gives

Question 9.5 How will you convert:
(iv) Ethanamine into methanamine
Answer :
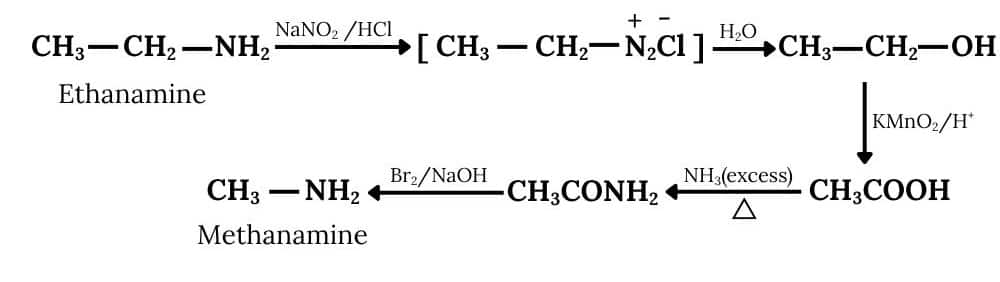
Ethanamine reacts with nitrous acid to form an azo compound, which further reacts with water to form ethanol and this on oxidation gives ethanoic acid. After treating with an excess of ammonia, the ethanoic acid becomes ethanamide, which further reacts with bromine and a strong base (Hoffmann bromamide degradation reaction), to form methanamine.
Question 9.5 How will you convert:
(v) Ethanoic acid into propanoic acid
Answer :
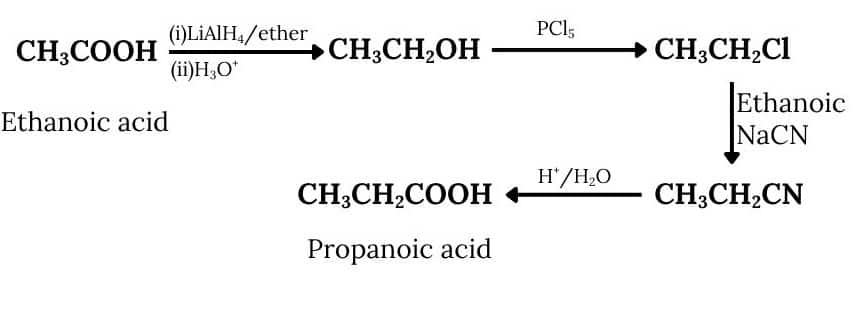
Acetic acid, on reduction with lithium aluminium hydride followed by acid hydrolysis, gives ethanol, which on reacting with
Question 9.5 How will you convert:
(vi) Methanamine into ethanamine
Answer :
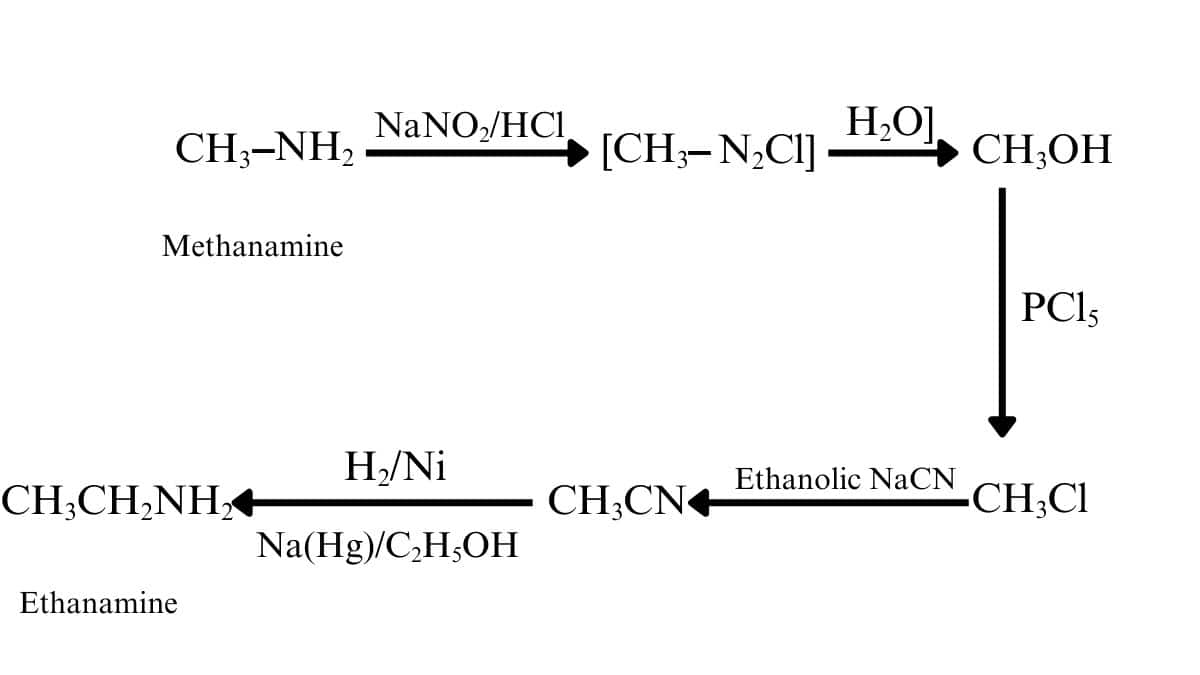
Methanamine on diazotisation gives methane diazonium salt, which on further hydrolysis forms methanol. Methanol on reacting with
Question 9.5 How will you convert:
(vii) Nitromethane into dimethylamine
Answer :
Conversion of Nitromethane into dimethylamine is shown below:
Reduction of Isocyanide
Question 9.5 How will you convert:
(viii) Propanoic acid into ethanoic acid?
Answer :
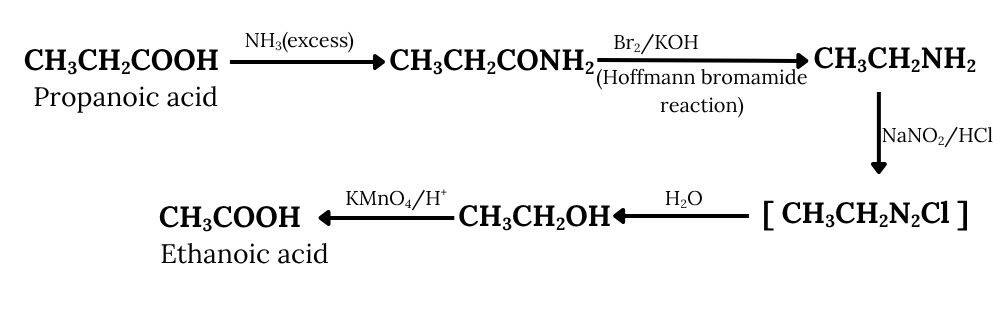
Propanoic acid on reacting with an excess of ammonia, gives propanamide, which further reacts with bromine and potassium hydroxide (Hoffmann bromamide degradation reaction) to give ethanamine. On diazotisation, ethanamine is converted into an azo salt, which on treatment with water forms ethanol. Ethanol on oxidation, provides ethanoic acid.
Answer :
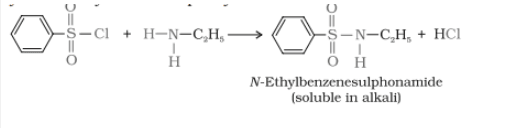
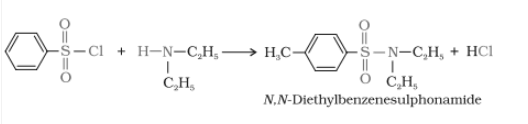
Primary, secondary, and tertiary amines are distinguished by Hinsberg's reagent test. In this test, the amines are allowed to react with benzene sulphonyl chloride (Hinsberg's reagent). All three types of amines give different products.
1.Primary amine reacts with benzene sulphonyl chloride
2. When secondary amines react, it gives N,N-diethylbenzene sulphonamide, which is insoluble in alkali.
3. Tertiary amine does not react with Hinsberg's reagent.
Question 9.7 Write short notes on the following:
Answer :
Carbylamine reaction-
Aliphatic and aromatic primary amines on reacting with chloroform and ethanolic KOH form isocyanides or carbylamine, which is a foul-smelling substance. Secondary and tertiary amines do not give this reaction. This reaction is known as the carbylamine reaction or isocyanide test. It is used to distinguish primary amines.
Question 9.7 Write short notes on the following:
Answer :
Aromatic primary amines react with nitrous acid (
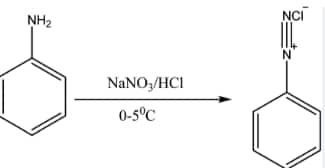
Question 9.7 Write short notes on the following:
(iii) Hofmann’s bromamide reaction
Answer :
Hofmann’s bromamide reaction-
When an amide is treated with bromine in aqueous solution or an ethanolic solution of sodium hydroxide, it produces primary amines with one less carbon atom than the parent compound. This reaction is known as the Hoffmann bromide degradation reaction.
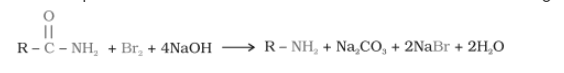
R = alkyl group like
Question 9.7 Write short notes on the following:
Answer :
The reaction of joining diazonium salt to the aromatic ring (like phenol) through a
Reactions-
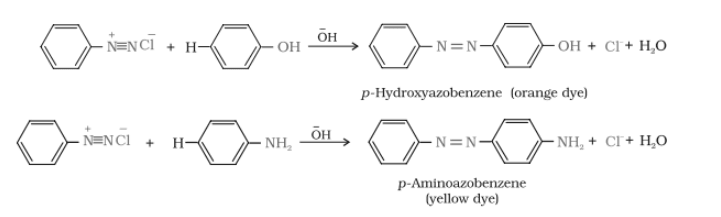
Diazonium salt reacts with aniline to give p-aminoazobenzene (yellow colour)
Question 9.7 Write short notes on the following:
Answer :
Ammonolysis-
When an alkyl or benzyl halide is going to react with the solution of ammonia(an ethanolic solution), it undergoes a nucleophilic substitution reaction in which the halogen atom is replaced by an amino (
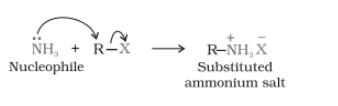
When this substituted ammonium salt is treated with a strong base, it gives a primary amine
Question 9.7 Write short notes on the following:
Answer :
Acetylation-
The introduction of an acetyl group (
Example-
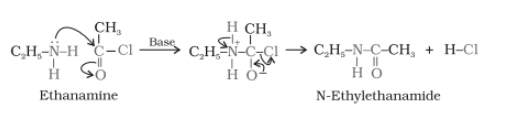
Aliphatic and aromatic primary or secondary amines undergo acetylation reaction when treated with acid chlorides, anhydrides, or esters by nucleophilic substitution. Here Hydrogen atom of
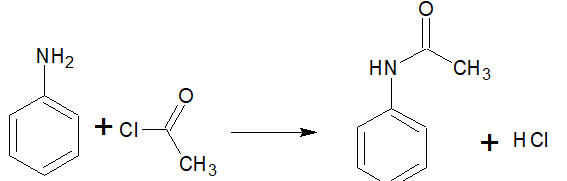
Question 9.7 Write short notes on the following:
(vii) Gabriel phthalimide synthesis.
Answer :
Gabriel phthalimide synthesis.-
It is mainly used for aliphatic primary amine preparation. Phthalimide reacts with ethanolic potassium hydroxide to produce the potassium salt of phthalimide. Upon heating with an alkyl halide and subsequent alkaline hydrolysis, this salt yields the corresponding primary amine. Aromatic amines cannot be produced by this method due to the inability of aryl halides to undergo nucleophilic substitution reaction with the anion produced by phthalimide.
Question 9.8 Accomplish the following conversions:
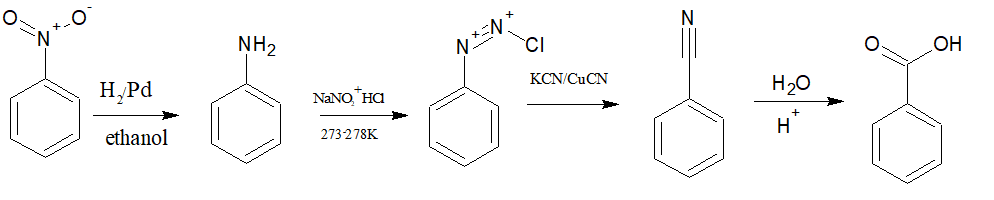
(i) Nitrobenzene to benzoic acid
Answer :
First, reduce nitrobenzene into aniline with the help of
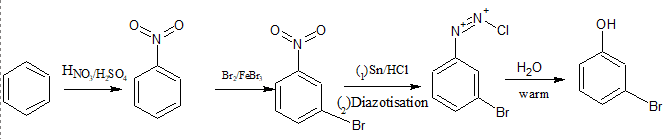
Question 9.8 Accomplish the following conversions:
Answer :
First, benzene is nitrated to form nitrobenzene, which is a meta-directing group and thus directs incoming substituents to the meta position. Next, bromination of nitrobenzene is carried out, introducing a bromine atom at the meta position relative to the nitro group.
The nitro group is then reduced to an amine using
Question 9.8 Accomplish the following conversions:
Answer :
On reacting benzoic acid with
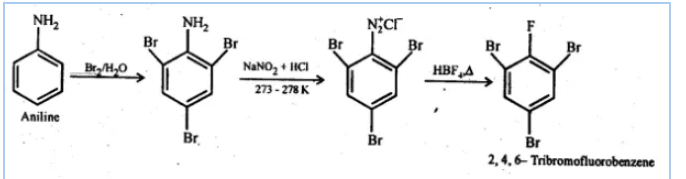
Question 9.8 Accomplish the following conversions:
(iv) Aniline to 2,4,6-tribromofluorobenzene
Answer :
First, we will do the bromination of aniline which gives 2,4,6-tribromoaniline. After that, we will perform diazotisation reaction which converts
Question 9.8 Accomplish the following conversions:
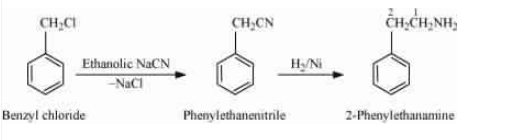
(v) Benzyl chloride to 2-phenylethanamine
Answer :
On reacting benzyl chloride with the ethanolic sodium cyanide (
Question 9.8 Accomplish the following conversions:
Answer :
On nitration of chlorobenzene, it gives p-nitrochlorobenzene, which is on further reaction with
Question 9.8 Accomplish the following conversions:
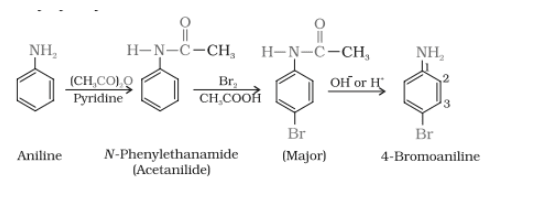
Answer :
Aniline on reacting with the acetic anhydride in the presence of pyridine, gives N-phenylethanamide, which on further reaction with
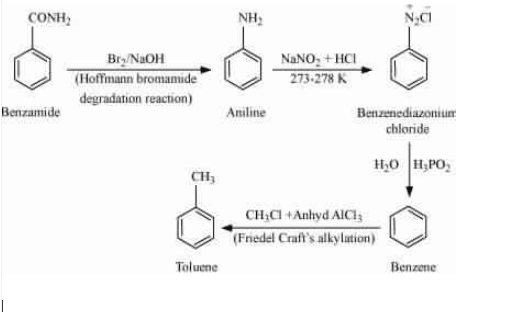
Question 9.8 Accomplish the following conversions:
Answer :
Use the Hoffmann bromamide degradation reaction so that it gives aniline and then do diazotisation reaction so that
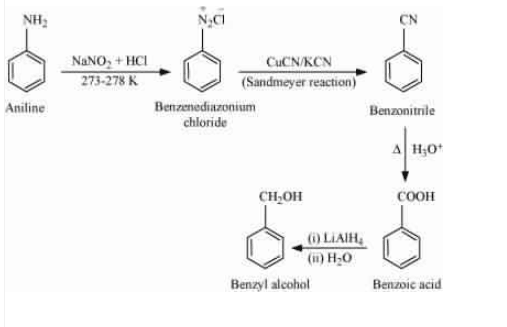
Question 9.8 Accomplish the following conversions:
(ix) Aniline to benzyl alcohol
Answer :
Aniline on treating with nitrous acid followed by sandmeyers reaction gives benzonitrile (
Question 9.9 Give the structures of A, B and C in the following reactions:
Answer :
In the first reaction, the iodine is replaced by CN, so the structure of (A) is
On partial hydrolysis, the CN converted in to
Question 9.9 Give the structures of A, B and C in the following reactions:
Answer :
The first part is the Sandmeyers reaction so CN replaces the
(C) benzamide
Question 9.9 Give the structures of A, B and C in the following reactions:
Answer :
The structure of A,B and C is
In the first reaction, the bromine is replaced by a cyanide molecule (
Question 9.9 Give the structures of A, B and C in the following reactions:
Answer :
A-
B-
C-
The first nitrobenzene reduced to aniline and then it reacts with
Question 9.9 Give the structures of A, B and C in the following reactions:
Answer :
A- ethanamide (
B- methenamine (
C- methanol (
Acetic acid reacts with ammonia, giving ethanamide and the second reaction is the Hoffmann bromamide degradation reaction, so it removes the -CO group and forms methanamine. The reaction with NaNO2/HCl gives methanol.
Question 9.9 Give the structures of A, B and C in the following reactions:
Answer :
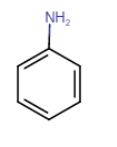
Initially, product A is the reduction of nitrobenzene means it is an aniline. When A react with
A= Aniline
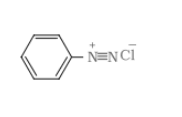
B = benzene diazonium chloride
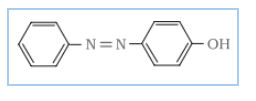
C= p-Hydroxyazobenzene
Answer :
The molecular formula of C =
It is a Hoffmann bromamide degradation reaction so the product should be
So B should be benzamide (
Therefore, the above reaction proceeds as
Question 9.11 Complete the following reactions:
Answer :
The reaction given above is carbylamine reaction in which primary aromatic amines react with chloroform and alcoholic potassium hydroxide to give isocyanide or carbylamine as a product, which is a foul-smelling substance.
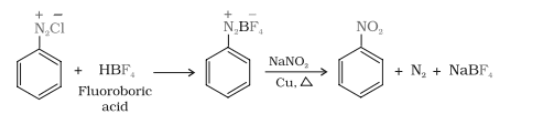
Question 9.11 Complete the following reactions:
Answer :
When benzene diazonium chloride reacts with
Question 9.11 Complete the following reactions:
Answer :
Aniline is a base and
Question 9.11 Complete the following reactions:
Answer :
Ethanol reduces the benzene diazonium chloride to benzene and oxidises into ethanal (
Question 9.11 Complete the following reactions:
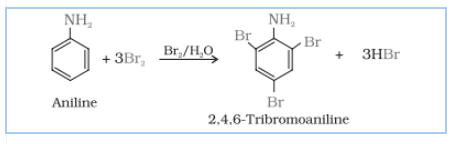
Answer :
Aniline on bromination in the presence of water at room temperature, gives a white precipitate of 2,4,6-tribromoaniline
Question 9.11 Complete the following reactions:
Answer :
Aniline reacts with acetic anhydride by a nucleophilic substitution reaction. Here, the H atom from
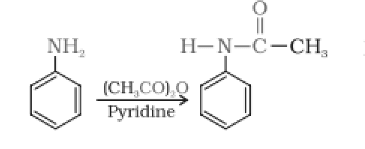
Question 9.11 Complete the following reactions:
Answer :
In this reaction, the chloride ion is replaced by
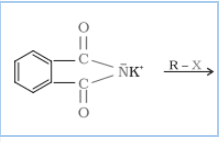
Question 9.12 Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Answer :
Gabriel phthalimide synthesis is mainly used for aliphatic primary amine preparation. Phthalimide reacts with ethanolic potassium hydroxide and produces the potassium salt of phthalimide, which on heating with alkyl halide, followed by alkaline hydrolysis, gives the corresponding primary amine.
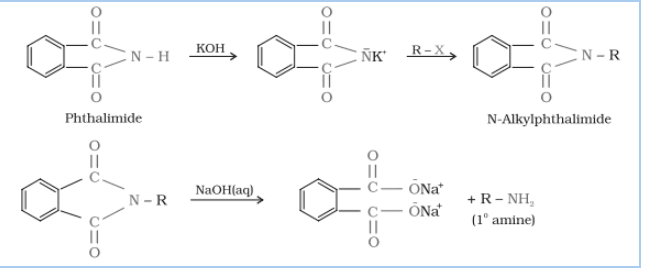
Aromatic amines cannot be produced by this method due to the inability of aryl halides to undergo nucleophilic substitution reaction with the anion produced by phthalimide.
Hence, aromatic amines cannot be produced by this method.
Question 9.13 Write the reactions of
(i) aromatic primary amines with nitrous acid.
Answer :
Aromatic primary amines react with nitrous acid(
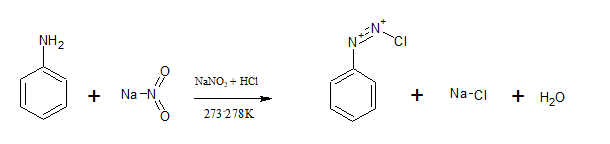
Question 9.13 Write the reactions of
(ii) aliphatic primary amines with nitrous acid.
Answer :
When aliphatic primary amines react with nitrous acid to form unstable diazonium salts, which on further reaction produce alcohol and hydrochloric acid (
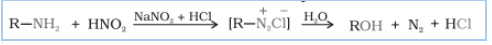
Question 9.14 Give a plausible explanation for each of the following:
(i) Why are amines less acidic than alcohols of comparable molecular masses?
Answer :
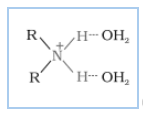
Protonation of amines gives-
Protonation of alcohol gives -
The negative charge is easily accommodated by the O atom because it is more electronegative than N atom. So, the amide ion is less stable than the alkoxide ion. Hence, amines are less acidic than alcohols.
Question 9.14 Give a plausible explanation for each of the following:
(ii) Why do primary amines have a higher boiling point than tertiary amines?
Answer :
The extent of intermolecular hydrogen bonding in primary amines is higher than that of secondary amines. Secondary amine has only one hydrogen, and tertiary amines have no hydrogen atom for intermolecular hydrogen bonding, whereas primary amine has two hydrogen atoms available for intermolecular hydrogen bonding. And we know that the higher the extent of hydrogen bonding, the higher is the boiling point.
In
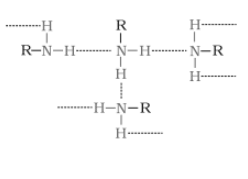
(In
(In
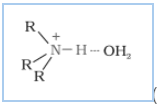
Question 9.14 Give plausible explanation for each of the following:
(iii) Why are aliphatic amines stronger bases than aromatic amines?
Answer :
Aliphatic amines are stronger bases than aromatic amines due to resonance. In aromatic amines, the lone pair of electrons at N atom is delocalised over the benzene ring, which makes it less available for electron donation. On the other hand, in aliphatic amines (
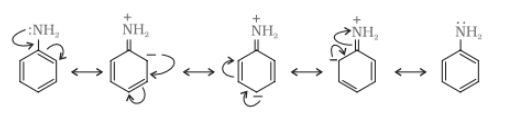
Practice the questions below to improve your problem-solving skills.
Question: When a concentrated solution of sulphanilic acid and 1-naphthylamine is treated with nitrous acid (273 K) and acidified with acetic acid, the mass (g) of 0.1 mole of product formed is : (Given molar mass in g mol–1 H : 1, C : 12, N : 14,O : 16, S : 32)
1) 343
2) 330
3) 33
4) 66
Answer:
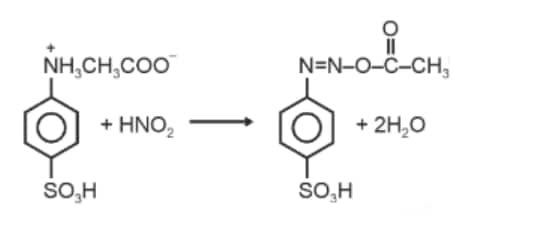
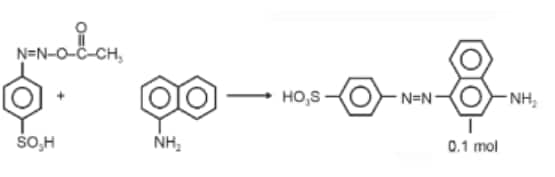
Molar mass of the product formed
Mass of 0.1 mol product
Hence, the correct answer is option (3).
Question: The major product (A) formed in the following reaction sequence is

1)

2)

3)

4)

Answer:

Hence, the correct answer is option (1).
Question:
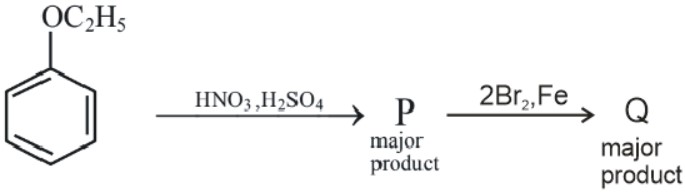
The ratio of the number of oxygen atoms to bromine atoms in the product
Answer:
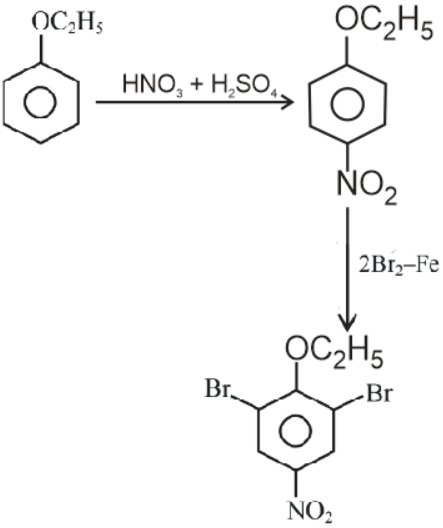
No. of oxygens/ No.of bromine atoms = 3/2 = 1.5
= 15 x 10-1.
Hence, the answer is (15).
To effectively solve questions of Amines, one should follow a structured approach. Here are some points that can help you with problem-solving strategies
Firstly, the understanding of basic concepts is necessary to solve the problems related to amines like
Take Aakash iACST and get instant scholarship on coaching programs.
Preparation methods are quite important from the exam point of view. Some of them includes,
Then, carefully study the physical properties of compounds like
Students can make use of short notes or charts to remember the concepts
Also, the test to distinguish the amines is too crucial, such as the Carbylamine test, Hinsberg's test and Nitrous acid reaction.
Solve the problems involving molecular weight, percentage yield and reaction stoichiometry. Also, solve the in-text and exercise problems provided in the NCERT textbook
The topics given in the chapter are
9.1 Structure of Amine
9.2 Classification
9.3 Nomenclature
9.5 Physical Properties
9.6 Chemical Properties
9.7 Method of Preparation of Diazoniun Salts
9.8 Physical Properties
9.9 Chemical Reactions
9.10 Importance of Diazonium Salts in the Synthesis of Aromatic Compounds
The links below will help you classify the topics on the basis of exams and prepare accordingly.
The NCERT solutions for the other chapters have also been designed. Click on the links below-
Excel your preparation by solving the NCERT solutions for other subjects as well.
Follow the links below to get your hands on the NCERT exemplar for other subjects for better learning.
| NCERT Exemplar Class 12th Maths Solutions |
| NCERT Exemplar Class 12th Physics Solutions |
| NCERT Exemplar Class 12th Chemistry Solutions |
| NCERT Exemplar Class 12th Biology Solutions |
Get the syllabus and books from the links provided in the table below
| NCERT Books Class 12 Chemistry |
| NCERT Syllabus Class 12 Chemistry |
| NCERT Books Class 12 |
| NCERT Syllabus Class 12 |
Amines are organic compounds derived from ammonia by replacing one or more hydrogen atoms with alkyl or aryl groups. They are important because they form the basis of many biologically active compounds, including amino acids, proteins, vitamins and neurotransmitters.
A quaternary ammonium salt is formed when the nitrogen atom in an amine is bonded to four alkyl or aryl groups and carries a positive charge. They are salts and therefore are often water soluble.
Ammonolysis yields a mixture of primary, secondary and tertiary amines and also quaternary ammonium salts. This is because the amine formed in the first step can further react with the alkyl halide. This issue is addressed to some extent by using a large excess of ammonia which favors the formation of the primary amine.
The Gabriel synthesis produces pure primary amines without contamination by secondary or tertiary amines.
The carbylamine test is a chemical test used to detect primary amines both aliphatic and aromatic. When heated with chloroform and alcoholic KOH , primary amines produce a foul-smelling isocyanide. Secondary and tertiary amines do not give this test.
Application Date:24 July,2025 - 23 August,2025
Application Date:24 July,2025 - 23 August,2025
Changing from the CBSE board to the Odisha CHSE in Class 12 is generally difficult and often not ideal due to differences in syllabi and examination structures. Most boards, including Odisha CHSE , do not recommend switching in the final year of schooling. It is crucial to consult both CBSE and Odisha CHSE authorities for specific policies, but making such a change earlier is advisable to prevent academic complications.
Hello there! Thanks for reaching out to us at Careers360.
Ah, you're looking for CBSE quarterly question papers for mathematics, right? Those can be super helpful for exam prep.
Unfortunately, CBSE doesn't officially release quarterly papers - they mainly put out sample papers and previous years' board exam papers. But don't worry, there are still some good options to help you practice!
Have you checked out the CBSE sample papers on their official website? Those are usually pretty close to the actual exam format. You could also look into previous years' board exam papers - they're great for getting a feel for the types of questions that might come up.
If you're after more practice material, some textbook publishers release their own mock papers which can be useful too.
Let me know if you need any other tips for your math prep. Good luck with your studies!
It's understandable to feel disheartened after facing a compartment exam, especially when you've invested significant effort. However, it's important to remember that setbacks are a part of life, and they can be opportunities for growth.
Possible steps:
Re-evaluate Your Study Strategies:
Consider Professional Help:
Explore Alternative Options:
Focus on NEET 2025 Preparation:
Seek Support:
Remember: This is a temporary setback. With the right approach and perseverance, you can overcome this challenge and achieve your goals.
I hope this information helps you.
Hi,
Qualifications:
Age: As of the last registration date, you must be between the ages of 16 and 40.
Qualification: You must have graduated from an accredited board or at least passed the tenth grade. Higher qualifications are also accepted, such as a diploma, postgraduate degree, graduation, or 11th or 12th grade.
How to Apply:
Get the Medhavi app by visiting the Google Play Store.
Register: In the app, create an account.
Examine Notification: Examine the comprehensive notification on the scholarship examination.
Sign up to Take the Test: Finish the app's registration process.
Examine: The Medhavi app allows you to take the exam from the comfort of your home.
Get Results: In just two days, the results are made public.
Verification of Documents: Provide the required paperwork and bank account information for validation.
Get Scholarship: Following a successful verification process, the scholarship will be given. You need to have at least passed the 10th grade/matriculation scholarship amount will be transferred directly to your bank account.
Scholarship Details:
Type A: For candidates scoring 60% or above in the exam.
Type B: For candidates scoring between 50% and 60%.
Type C: For candidates scoring between 40% and 50%.
Cash Scholarship:
Scholarships can range from Rs. 2,000 to Rs. 18,000 per month, depending on the marks obtained and the type of scholarship exam (SAKSHAM, SWABHIMAN, SAMADHAN, etc.).
Since you already have a 12th grade qualification with 84%, you meet the qualification criteria and are eligible to apply for the Medhavi Scholarship exam. Make sure to prepare well for the exam to maximize your chances of receiving a higher scholarship.
Hope you find this useful!
hello mahima,
If you have uploaded screenshot of your 12th board result taken from CBSE official website,there won,t be a problem with that.If the screenshot that you have uploaded is clear and legible. It should display your name, roll number, marks obtained, and any other relevant details in a readable forma.ALSO, the screenshot clearly show it is from the official CBSE results portal.
hope this helps.

Take Aakash iACST and get instant scholarship on coaching programs.

This ebook serves as a valuable study guide for NEET 2025 exam.

This e-book offers NEET PYQ and serves as an indispensable NEET study material.

As per latest syllabus. Physics formulas, equations, & laws of class 11 & 12th chapters
As per latest syllabus. Chemistry formulas, equations, & laws of class 11 & 12th chapters
As per latest 2024 syllabus. Study 40% syllabus and score upto 100% marks in JEE
We endeavor to keep you informed and help you choose the right Career path. Sign in and access our resources on Exams, Study Material, Counseling, Colleges etc.