NCERT Solutions for Class 9 Science Chapter 3 - Tissue in Action
NCERT Solution for Class 9 Science Chapter 3 Tissue in Action gives detailed answers to all Revise, Reflect, Refine exercises and in-text questions. It includes why plants have meristematic tissues for growth? How do types of animal tissues like epithelial, connective, muscular, and nervous tissues work together? The answer is explained in this tissue chapter 3 solution. Students can download the Exploration NCERT Chapter 3 tissue solution PDF for structured study and quick revision.
This Story also Contains
- NCERT Solutions for Class 9 Science Chapter 3 Tissue in Action - Download PDF
- Access Revise, Reflect, Refine Exercise Questions With Solutions of Class 9 Science Chapter 3 Tissues
- Get Class 9 Science Chapter 3 Tissue (In-text Questions): Think It Over and Pause and Ponder Answers
- Approach to Solve Tissues Class 9 Question Answer
- Important Topics of Class 9 NCERT Science Chapter 3 Tissues
- What Will You Learn From Tissues NCERT Solutions?
- Why Class 9 Science Chapter 3 Tissues NCERT Solutions are Important?

The Class 9 Science chapter 3 tissue NCERT solutions PDF includes important questions such as meristematic vs permanent plant tissues, and which specific function they perform. It includes parenchyma, collenchyma, and sclerenchyma, and vascular tissue like xylem and phloem. This article includes updated NCERT Exploration questions of Revise, Reflect, Refine exercise step-by-step answers, diagrams, and a PDF download option for direct access to the answers.
NCERT Solutions for Class 9 Science Chapter 3 Tissue in Action - Download PDF
Students can download the complete questions with detailed answers for better learning. Tissues Class 9 questions and answers PDF can be accessed offline without needing the internet. All the answers in the NCERT Solutions for Class 9 are presented in easy and simple language.
Access Revise, Reflect, Refine Exercise Questions With Solutions of Class 9 Science Chapter 3 Tissues
The detailed answers to all the Revise, Reflect, Refine questions in this chapter are given below. Students learn about the different types of tissues in plants and animals and what role they play. Doing regular practice of the Class 9 Science Chapter 3 Tissues question answer is important to score well.
Revise, Reflect, Refine Exercise
Question 1: Meristematic tissues divide repeatedly. What property of their cells allows them to do this?
(i) They have thick walls for protection.
(ii) They contain large vacuoles that store nutrients.
(iii) They have thin walls, dense cytoplasm and a large, prominent nucleus.
(iv) They are functionally differentiated cells.
Answer: The correct option is (iii) They have thin walls, dense cytoplasm and a large, prominent nucleus.
Explanation: Meristematic cells are young and actively dividing. They have thin cell walls, so they can grow easily. They have dense cytoplasm to support metabolism, and a large nucleus that controls cell division. They lack large vacuoles and permanent differentiation. This keeps them in a dividing state and helps plants grow continuously.
Question 2: If a plant is unable to transport food from leaves to roots, which tissue is malfunctioning?
(i) Xylem
(ii) Phloem
(iii) Epidermis
(iv) Sclerenchyma
Answer: The correct option is (ii) Phloem
Explanation: Plants make food in their leaves by photosynthesis.
This food must reach roots, stems, and other parts. Phloem carries food made in leaves to other parts of the plant. If phloem does not work, food cannot reach the roots and stem. This results in improper roots and other parts' growth.
Question 3: Why are the epithelial tissues that line an animal’s internal organs usually only one or a few cells thick?
(i) To store food efficiently.
(ii) To provide maximum strength.
(iii) To allow quick exchange of materials across them.
(iv) To reduce friction
Answer: The correct option is (iii) To allow quick exchange of materials across them.
Explanation: Epithelial tissues cover and protect internal organs. They are thin and usually have one or a few cells thick. This thinness helps gases, nutrients, and waste move quickly. For example, in the lungs, oxygen passes easily through the thin epithelial lining. If the lining were thick, exchange would become slow, and organs would not work well.
Question 4: You can perform these two jumps given in Fig.
Straight-leg jump — keep knees and ankles stiff.
Normal jump — bend knees and ankles naturally.
How did your ankle, knee and hip positions differ between the two jumps?

Answer:
|
Jump Type |
Ankle Position |
Knee Position |
Hip Position |
Effect on Movement |
|
Straight‑leg jump |
Stiff, no bending |
Stiff, locked |
Rigid, less flexible |
Harder jump, poor balance, and landing is uncomfortable |
|
Normal jump |
Flexed, bends naturally |
Bent, absorbs shock |
Free, moves easily |
Smooth jump, better balance, higher and safer movement |
Question 5: Which type of joint is involved when you bend your knees and ankles?
(i) Ball and socket
(ii) Hinge
(iii) Pivot
Answer: The correct answer is (ii) Hinge
The joint present in the knees and ankles is the hinge joint. It allows movement in one direction, just like a door hinge. Knees bend forward and backwards only. Ankles also bend mainly in one direction.
(i) They cannot rotate freely like a ball-and-socket joint (shoulder, hip).
(iii) They also do not twist like a pivot joint (neck).
Question 6: In each of the following cases (A, B, C and D), choose the correct option as given below:
(i) Both (A) and (R) are true, and (R) is the correct explanation of (A).
(ii) Both (A) and (R) are true, but (R) is not the correct explanation of (A).
(iii) (A) is true, but (R) is false.
(iv) (A) is false, but (R) is true.
A. Assertion: Epithelium is well-suited for gas exchange in the lungs.
Reason: It consists of multiple layers of tall cells that slow down diffusion.
B. Assertion: Cardiac muscle can contract continuously without fatigue.
Reason: Cardiac muscle cells have a high number of mitochondria and an abundant blood supply.
C. Assertion: Tendons connect bone to bone and allow joint movement.
Reason: Tendons are made of tough connective tissue that transmits force from muscle to bone.
D. Assertion: In a hinge joint, movement occurs primarily in one plane.
Reason: The bone ends are shaped to allow sliding in all directions.
Answer:
A. Correct option: (iii) (A) is true, but (R) is false.
Epithelium in the lungs is thin and single‑layered. It helps fast gas exchange. The statement about multiple tall layers slowing diffusion is wrong.
B. Correct option: (i) Both (A) and (R) are true, and (R) is the correct explanation of (A).
Cardiac muscle contracts continuously without fatigue because it has many mitochondria and a rich blood supply. This gives constant energy and oxygen.
C. Correct option: (iv) (A) is false, but (R) is true.
Tendons connect muscle to bone, not bone to bone. Ligaments connect bone to bone. The reason for tendons transmitting force is correct.
D. Correct option: (iii) (A) is true, but (R) is false.
Hinge joints allow movement mainly in one plane, like knees and elbows. The idea of sliding in all directions is incorrect.
Question 7: Plot a graph between the age of a tree (in years) on the x-axis and the diameter of the tree (in cm), along with the number of annual rings formed over time on the y-axis, using the data given in the Table
Table: Data related to the age of a teak tree, and corresponding increase in the diameter of the stem and number of annual rings
|
S. No. |
Age of the teak tree (Years) |
DBH (Diameter at Breast Height) of tree (cm) |
Number of annual rings formed |
|
1. |
5 |
4 |
5 |
|
2. |
10 |
8 |
10 |
|
3. |
20 |
24 |
20 |
|
4. |
25 |
28 |
25 |
|
5. |
30 |
32 |
30 |
|
6. |
40 |
40 |
40 |
(i) Analyse the graph in terms of the diameter of the stem over time and share the interpretation.
(ii) What is the relation between the diameter of the teak tree to the annual rings formed?
(iii) Which specialised tissue is responsible for the girth of the stem and where is it located?
Answer:
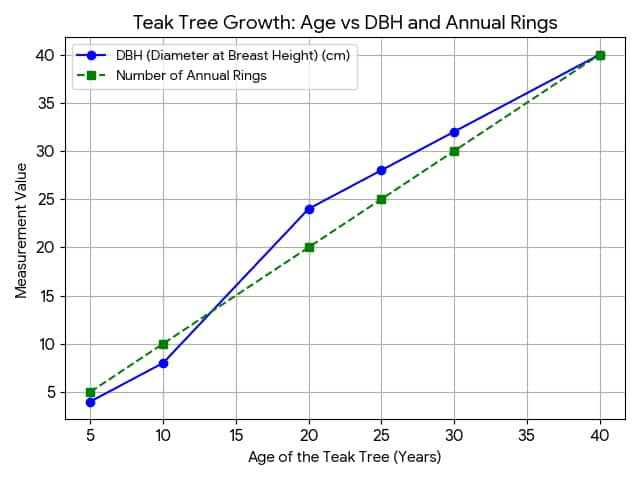
(i) The diameter of the teak tree increases with age at a non - uniform rate. Between 10 and 20 years, the diameter rises sharply from 8 cm to 24 cm, called a period of rapid growth. Growth becomes slower and steadier after 20 years. Overall, the tree shows continuous lateral growth throughout life.
(ii) The number of annual rings equals the tree’s age in years. As the annual rings increase, the diameter also increases. Each ring represents one year of growth by the lateral meristem.
(iii) The specialised tissue responsible for girth is the lateral meristem. It lies between xylem and phloem. It forms a ring around the stem. The cambium divides repeatedly to produce secondary xylem and phloem, increasing the girth of the stem. Each year, it adds a new ring of wood and forms the annual growth rings.
Question 8: In a forest, it was observed that one of the trees was severely debarked by an elephant to meet its food requirements, as the bark is a rich source of nutrients (Fig.). Based on your learning, answer the following:

(i) Which function(s) of the tree is/are hampered by debarking?
(ii) Which plant tissue would be affected by further damage to the tree trunk even after debarking?
(iii) Which function of the tree would be hampered if the tissues beneath the bark were severely damaged?
(iv) What assumptions are you making to answer the questions above? How would the answer change if your assumptions are also changed?
Answer:
(i) The functions of the tree which are hampered by debarking are protection, food transport, and water loss. Bark protects the tree from pests and weather. Phloem in bark carries food from leaves to roots. Removing bark also increases water loss from exposed tissues.
(ii) The plant tissue affected by further damage after debarking is phloem, cambium, and xylem. Phloem carries food, cambium produces new cells for growth, and xylem transports water. If these tissues are harmed, the tree cannot survive.
(iii) The function hampered if tissues beneath the bark are damaged is water and mineral transport. The xylem lies under the bark. It carries water and minerals from roots to leaves. If xylem is destroyed, leaves dry, photosynthesis stops, and the tree dies.
(iv) The assumption is that the elephant removed the bark all around the trunk, called girdling. If this assumption changes and only a small patch is removed, the tree may survive because food and water transport continue through the remaining tissues.
Question 9: Aamrapali observed that a young mango sapling’s stem bends flexibly during monsoon winds and does not break. Which tissue is responsible for this flexibility? Predict and provide your explanation of the impact if the existing tissue was replaced by sclerenchyma.
Answer: The flexibility of the young mango sapling’s stem is due to collenchyma tissue. Collenchyma cells have unevenly thickened walls. They provide mechanical support while keeping the stem bendable during wind.
If collenchyma were replaced by sclerenchyma, the stem would become rigid and hard. Sclerenchyma cells are dead and have thick lignified walls. The sapling would lose flexibility, bend less, and break easily in strong winds.
Question 10: Sohan designed an experiment for the regeneration of sugarcane, where he used cuttings to grow sugarcane. He used two types of cuttings, type ‘A’ and type ‘B’ (Fig.). After a few weeks, type ‘B’ cuttings sprouted and developed into sugarcane plants, whereas the type ‘A’ cuttings did not sprout.

(i) Why were the type ‘B’ cuttings able to grow as sugarcane but type ‘A’ could not?
(ii) What difference was present in type ‘B’ compared to type ‘A’?
(iii) What observation or measurement was made to determine whether this change had an effect?
(iv) What parameters should be kept the same for both types of cuttings to ensure a fair comparison?
Answer:
(i) Type B cuttings were able to grow into sugarcane plants because they had nodes and buds. These structures allow new shoots and roots to develop. Type A cuttings lacked nodes, so they could not sprout or regenerate.
(ii) The main difference between Type B and Type A cuttings was the presence of nodes with buds. Type B had these nodes essential for regeneration. Type A did not have nodes, so no new shoots could form.
(iii) The observation made was the sprouting of buds from the cuttings. The measurement was the growth of new shoots and roots. This showed that Type B cuttings regenerated successfully, while Type A cuttings failed to grow.
(iv) To assure a fair comparison, both cuttings must be planted under the same conditions.
-
Soil type must be the same.
-
The water supply must be the same.
-
Sunlight must be the same.
-
The temperature must be the same.
-
Planting depth must be the same.
This ensures that only the presence of nodes affects growth.
Question 11: During the discussion in class, Rohan gives a statement that, “A tissue is a group of similar cells performing similar functions”. But Rajiv counter argues that, “this is true in case of simple tissues but little different in case of complex tissues”. Provide your explanation in view of the discussion in class.
Answer: Rohan said that a tissue is a group of similar cells performing similar functions. This is true for simple tissues like parenchyma, collenchyma, and sclerenchyma.
Rajiv explained that complex tissues are different. Complex tissues like xylem and phloem have different types of cells. Each cell performs a different role, but together they complete one main function.
For example, xylem has tracheids, vessels, fibres and parenchyma. Tracheids and vessels transport water, fibres give strength and parenchyma stores food. Phloem has sieve tubes, companion cells, fibres and parenchyma. They all work together to transport food.
So, both Rohan and Rajiv are correct in different ways.
Question 12: Coconut husk fibres are used for mats which are tough and fibrous. Which tissue has structural features suitable for providing this strength? Explain why living parenchyma couldn’t serve the same purpose.
Answer: Coconut husk fibres are strong and tough because they are made of sclerenchyma tissue. Sclerenchyma cells are dead and have very thick lignified walls. This makes them hard and durable. That is why they are used to make mats, ropes and brushes. Living parenchyma could not serve this purpose.
Parenchyma cells are soft, thin-walled, and living. They store food and water, but they cannot provide strength. If mats were made of parenchyma, they would break easily and not last long.
Hence, sclerenchyma is the correct tissue for strength because its structure is designed to resist pressure and provide support.
Question 13: Vibha claims to her friend Neha that, “Meristematic cells are located only at the root and shoot apices”. What do you think about this statement? What question can Neha ask Vibha to help her understand further if the statement is incorrect?
Answer: Vibha said that meristematic cells are located only at the root and shoot apices. This is partly correct but incomplete.
Reason: Meristematic cells are also found in other regions of the plant. They are present in the lateral meristem intercalary meristem. There are different types of meristematic tissues at different locations and functions.
|
Type of Meristem |
Location |
Function |
|
Apical meristem |
Root tips and shoot tips |
Increases the length of roots and shoots |
|
Lateral meristem |
Along the sides of stems |
Increases the girth and thickness of the stem |
|
Intercalary meristem |
Base of leaves and nodes |
Helps regrowth after cutting or grazing |
Neha can ask Vibha questions like:
-
If meristems are only at the tips?
-
How does a tree trunk grow thicker every year?
-
How does grass grow back after mowing or grazing if the tip is removed?
These questions show that meristematic cells exist in more than one location and perform different growth functions.
Question 14: A plant cell and an animal cell are of the same size.
(i) Which cell will have a larger vacuole? Give reasons.
(ii) What assumptions are you making to answer the question above?
Answer:
(i) The plant cell will have a larger vacuole.
-
Plant vacuoles store water, minerals and waste.
-
They also maintain turgor pressure, which keeps the plant upright and firm.
-
Animal cells have only small vacuoles because they do not need large storage or pressure maintenance.
(ii) The assumptions are
-
Both cells are of the same size, healthy and living.
-
Comparison is made under normal conditions without any disease or abnormal growth.
Question 15. A textbook states, “Each plant tissue performs only one specific function”. What questions would you ask to critically examine the correctness of this statement? What examples of tissues would you take to find out the answers to these questions?
Answer: The textbook statement that each plant tissue performs only one specific function is not fully correct. To examine this critically, we can ask questions like:
-
Can a tissue perform more than one role?
-
Do tissues sometimes combine storage with support or transport?
Examples -
-
Xylem transports water but also provides mechanical strength through fibres.
-
Phloem transports food, but also stores it and gives support.
-
Parenchyma stores food, but when it contains chlorophyll can also perform photosynthesis.
-
Even collenchyma provides support and flexibility at the same time.
Thus, plant tissues often perform multiple functions. The statement is oversimplified and should be refined to show that tissues may have one main function but can also contribute to other roles in the plant body.
Confused between CGPA and Percentage?
Get your results instantly with our calculator!
Get Class 9 Science Chapter 3 Tissue (In-text Questions): Think It Over and Pause and Ponder Answers
The detailed solution to all these in-text exercises in this chapter is given below. Studying the NCERT Solutions Class 9 Science Chapter 3 Tissue Think It Over and Pause & Ponder questions helps students to explore how tissue differences in plants and animals, types of joints involved in human beings' movement.
Think It Over
Question 1: How is the study of cells and tissues significant for understanding the life processes and human welfare?
Answer: The study of cells and tissues is significant because it helps us understand how life processes occur in plants and animals. It explains how different organs function and how growth and development take place. This knowledge is useful for human welfare in medicine, agriculture, and biotechnology.
Question 2: How are tissues in plants and animals different, and why?
Answer: Plant tissues and animal tissues are different because plants are fixed in one place, while animals can move. Plant cells have a rigid cell wall that provides support and allows photosynthesis. Animal cells lack a cell wall, and this makes them flexible and suitable for locomotion. Their tissues are adapted for digestion, movement, and nervous control.
Question 3: How is the division of labour at various levels of organisation in multicellular organisms correlated with their structure and function?
Answer: In multicellular organisms, division of labour means that different cells and tissues perform different functions. This is correlated with their structure. Muscle cells are elongated to contract and cause movement. Nerve cells are branched to transmit signals. Xylem vessels are tubular to conduct water. This specialisation makes life processes efficient.
Pause and Ponder
Page 33
Question 1: You may have noticed that fibres of coconut husk are hard and brittle, whereas the leaf stalks of coriander are soft and flexible. Find out the reason.
Answer: Fibres of coconut husk are hard and brittle because they are made of sclerenchyma tissue with thick lignified walls and dead cells. The leaf stalks of coriander are soft and flexible because they contain collenchyma tissue with living cells and unevenly thickened walls that provide flexibility.
Page 34
Question 2: Why do you think that a thick cuticle on the outer wall of epidermis is advantageous for a plant living in the desert but disadvantageous for a plant living underwater?
Answer: A thick cuticle on the epidermis is advantageous for desert plants because it reduces water loss and protects against heat. It is disadvantageous for underwater plants because it prevents easy exchange of gases and water absorption, which are essential in aquatic conditions.
Question 3: Once water is absorbed by plant roots, it has to travel against gravity through xylem. How do the ‘dead’ cells of the xylem work together with the living cells of leaves at the top to keep the water moving?
Answer: Dead xylem cells form hollow tubes that allow water to move upward by capillary action and transpiration pull. Living cells in leaves, through transpiration, create a suction that keeps water moving against gravity. Together, dead xylem cells and living leaf cells maintain continuous water transport.
Question 4: What do you think will happen if there were no stomata in the epidermis of the stem or leaves?
Answer: If there were no stomata in the epidermis of stems or leaves, gaseous exchange would not occur. Photosynthesis would stop due to a lack of carbon dioxide. Transpiration would also stop, preventing water movement in the xylem and disturbing the plant’s survival.
Page 40
Question 5: Look at the picture given below (Fig. ). Carefully observe the various poses of classical and folk dances of India. Can you identify which joints are involved? Also, what type of movement does each joint allow?

Answer: There are different joints of the body involved in classical and folk dance poses
- The ball-and-socket joint in the shoulder and hip allows free circular movement of arms and legs.
-
The hinge joint in the knee and elbow allows bending and straightening movements.
-
The pivot joint in the neck allows rotation of the head.
-
The gliding joint in the wrist and ankle allows sliding and flexible movements.
Each joint type enables specific dance movements and makes the poses expressive and graceful.
Approach to Solve Tissues Class 9 Question Answer
Provided below are a few useful tips that students can use to answer the questions of the tissue chapter effectively.
-
Students should start by understanding the classification of plant and animal tissues with examples.
-
Focus should be given to diagrams, labelling, and drawing tissues as they are frequently asked in the exam.
-
They should focus on the tissues' structure, their functions, and their locations in the body. Students can make flashcards of different tissues by using the Class 9 Science Tissues question answer.
-
The NCERT Solutions for Class 9 Science have all the solved exercise questions with the necessary information.
Important Topics of Class 9 NCERT Science Chapter 3 Tissues
The important topics of the tissues chapter are listed below. Knowing the main topics makes the preparation organised and systematic. The NCERT Solutions for Class 9 Science Chapter 3 Tissues include questions related to all these topics.
-
Plant Tissues
-
Muscular Tissue
-
Nervous Tissue
-
Skeletal System
-
Musculoskeletal System
What Will You Learn From Tissues NCERT Solutions?
Living beings are made up of groups of cells that perform specific tasks together. Students, by going through these solutions, learn a lot of interesting things, which are given below:
-
Students will learn the difference between plant and animal tissues.
-
The Tissues Class 9 question answer describes the role of xylem and phloem in plants.
-
They will understand the structure and function of meristematic and permanent tissues through clear examples.
-
They will gain knowledge about different types of tissues, such as muscle tissue, connective tissue, and nervous tissue.
-
Students will explore how tissues provide strength, support, and coordination in multicellular organisms through the Class 9 Science Tissues question answer.
Why Class 9 Science Chapter 3 Tissues NCERT Solutions are Important?
The chapter introduces students to the different types of plant and animal tissues, their structure, and the functions they play. Students studying the Tissues chapter will understand the structure and function of living organisms.
-
Class 9 Science Chapter 3 Tissues NCERT Solutions are important as they introduce plant and animal tissues.
-
It explains the structure and function relationship in tissues, helping students understand how organisms perform various activities.
-
The chapter builds the base for Class 11 and 12 topics in Human Anatomy, Plant Anatomy, and Physiology.
-
Understanding tissue structure and function is important as questions are frequently asked in the NEET and other competitive exams.
Frequently Asked Questions (FAQs)
A tissue is a group of similar cells that perform a specific function. In plants, tissues like xylem and phloem help in transport. In animals, tissues like epithelial, connective, muscular, and nervous perform different roles.
- Epithelial tissue – covers body surfaces.
- Connective tissue – supports and binds (bone, cartilage, blood).
- Muscular tissue – helps in movement.
- Nervous tissue – transmits signals.
You can find the NCERT Solutions PDF for Class 9 Science Chapter 3 – Tissues directly on Careers360. It includes step-by-step answers to the Revise, Reflect, Refine exercises, in-text questions, and important diagrams for exam preparation. The PDF is available for free download and can be accessed offline for revision.
The NCERT Solutions for Chapter 3 Tissues explain the difference between plant tissues and animal tissues. Students learn about meristematic vs permanent tissues, including parenchyma, collenchyma, sclerenchyma, xylem, and phloem. The chapter also covers animal tissues like epithelial, connective, muscular, and nervous tissues, along with the skeletal and musculoskeletal system.
Popular Questions
Courses After 12th
Applications for Admissions are open.
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Victoria University, Delhi NCR
ApplyApply for UG & PG programmes from Victoria University, Delhi NCR Campus
Illinois Tech Mumbai
ApplyAdmissions open for UG & PG programs at Illinois Tech Mumbai
University of Aberdeen Mumbai
ApplyApply for UG & PG courses at University of Aberdeen, Mumbai Campus
University of York, Mumbai
ApplyUG & PG Admissions open for CS/AI/Business/Economics & other programmes.
University of Bristol, Mumbai Enterprise Campus
ApplyBristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes

