University of Liverpool, Bengaluru Campus
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
The Central Board of Secondary Education has released the CBSE 11th Chemistry paper syllabus PDF for the 2025-26 academic session. The CBSE Class 11 Chemistry syllabus 2025-26 PDF is available at cbseacademic.nic.in. Students can also find the direct download link to the syllabus in the article below.
This Story also Contains

The CBSE Chemistry theory paper of Class 11 will carry 70 marks weightage, and 30 marks will be reserved for practicals and internal assessment. The CBSE Class 11 Chemistry syllabus for 2025-26 covers nine units. Main topics include Basic Concepts, Atomic Structure, Periodic Classification, Bonding, Thermodynamics, Equilibrium, and Redox Reactions. Organic Chemistry and Hydrocarbons carry the highest weightage together, contributing 21 marks.
Subject Name | PDF Download Link |
CBSE Class 11 Chemistry Syllabus 2025-26 |
Let us first have a look at the topics which will be covered in the CBSE Class 11 Chemistry theory paper. Students can also go through the marks allocation in the table below:
S. No | UNIT | Marks |
1 | Some Basic Concepts of Chemistry | 7 |
2 | Structure of Atom | 9 |
3 | Classification of Elements and Periodicity in Properties | 6 |
4 | Chemical Bonding and Molecular Structure | 7 |
5 | Chemical Thermodynamics | 9 |
6 | Equilibrium | 7 |
7 | Redox Reactions | 4 |
8 | Organic Chemistry: Some Basic Principles and Techniques | 11 |
9 | Hydrocarbons | 10 |
Total | 70 | |
In the following table, students can find the detailed unit-wise CBSE 11th Chemistry syllabus topics. Go through the table carefully and prepare accordingly for the final exams.
Unit | Topics covered |
Unit 1: Some Basic Concepts of Chemistry | General Introduction: Importance and scope of Chemistry, Nature of matter, laws of chemical combination, Dalton's atomic theory: concept of elements, atoms and molecules, atomic and molecular masses, mole concept and molar mass, percentage composition, empirical and molecular formula, chemical reactions, stoichiometry and calculations based on stoichiometry. |
Unit 2: Structure of Atom | Discovery of Electron, Proton and Neutron, atomic number, isotopes and isobars. Thomson's model and its limitations. Rutherford's model and its limitations, Bohr's model and its limitations, concept of shells and subshells, dual nature of matter and light, de Broglie's relationship, Heisenberg uncertainty principle, concept of orbitals, quantum numbers, shapes of s, p and d orbitals, rules for filling electrons in orbitals - Aufbau principle, Pauli's exclusion principle and Hund's rule, electronic configuration of atoms, stability of half-filled and completely filled orbitals. |
Unit 3: Classification of Elements and Periodicity in Properties | Significance of classification, brief history of the development of periodic table, modern periodic law and the present form of periodic table, periodic trends in properties of elements -atomic radii, ionic radii, inert gas radii, Ionization enthalpy, electron gain enthalpy, electronegativity, valiancy, Nomenclature of elements with atomic number greater than 100. |
Unit 4: Chemical Bonding and Molecular Structure | Valence electrons, ionic bond, covalent bond, bond parameters, Lewis structure, polar character of covalent bond, covalent character of ionic bond, valence bond theory, resonance, geometry of covalent molecules, VSEPR theory, concept of hybridization, involving s, p and d orbitals and shapes of some simple molecules, molecular orbital theory of homonuclear diatomic molecules (qualitative idea only), Hydrogen bond. |
Unit 5: Chemical Thermodynamics | Concepts of System and types of systems, surroundings, work, heat, energy, extensive and intensive properties, state functions. First law of thermodynamics -internal energy and enthalpy, heat capacity and specific heat, measurement of ∆U and ∆H, Hess's law of constant heat summation, enthalpy of bond dissociation, combustion, formation, atomization, sublimation, phase transition, ionization, solution and dilution. Second law of Thermodynamics (brief introduction), Introduction of entropy as a state function, Gibb's energy change for spontaneous and non- spontaneous processes, criteria for equilibrium, Third law of thermodynamics (brief introduction). |
Unit 6: Equilibrium | Equilibrium in physical and chemical processes, dynamic nature of equilibrium, law of mass action, equilibrium constant, factors affecting equilibrium – Le Chatelier's principle, ionic equilibrium- ionization of acids and bases, strong and weak electrolytes, degree of ionization, ionization of poly basic acids, acid strength, concept of pH, hydrolysis of salts (elementary idea), buffer solution, Henderson Equation, solubility product, common ion effect (with illustrative examples). |
Unit 7: Redox Reactions | Concept of oxidation and reduction, redox reactions, oxidation number, balancing redox reactions, in terms of loss and gain of electrons and change in oxidation number, applications of redox reactions. |
Unit 8: Organic Chemistry – Some Basic Principles and Techniques | General introduction, methods of purification, qualitative and quantitative analysis, classification and IUPAC nomenclature of organic compounds. Electronic displacements in a covalent bond: inductive effect, electrometric effect, resonance and hyper conjugation. Homolytic and heterolytic fission of a covalent bond: free radicals, carbocations, carbanions, electrophiles and nucleophiles, types of organic reactions. |
Unit 9: Hydrocarbons | Aliphatic Hydrocarbons Alkanes - Nomenclature, isomerism, conformation (ethane only), physical properties, chemical reactions including free radical mechanism of halogenation, combustion and pyrolysis. Alkenes - Nomenclature, structure of double bond (ethene), geometrical isomerism, physical properties, methods of preparation, chemical reactions: addition of hydrogen, halogen, water, hydrogen halides (Markovnikov's addition and peroxide effect), ozonolysis, oxidation, mechanism of electrophilic addition. Alkynes - Nomenclature, structure of triple bond (ethyne), physical properties, methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of - hydrogen, halogens, hydrogen halides and water. Aromatic Hydrocarbons Introduction, IUPAC nomenclature, benzene: resonance, aromaticity, chemical properties: mechanism of electrophilic substitution. Nitration, sulphonation, halogenation, Friedel-Craft's alkylation and acylation, directive influence of functional group in mono-substituted benzene, carcinogenicity and toxicity |
Get your results instantly with our calculator!
In the following table, students can find the CBSE Class 11 Chemistry syllabus for practicals. Check it out:
Section | Content |
A. Basic Laboratory Techniques | 1. Cutting glass tube and a glass rod 2. Bending a glass tube 3. Drawing out a glass jet 4. Boring a cork |
B. Characterisation and Purification of Chemical Substances | 1. Determination of the melting point of an organic compound 2. Determination of the boiling point of an organic compound 3. Crystallisation of an impure sample (choose one): Alum, Copper Sulphate, Benzoic Acid |
C. Experiments Based on pH | 1. Any one experiment: – Determination of pH of solutions from fruit juices or known concentrations using pH paper/universal indicator – Comparing the pH of strong and weak acids of the same concentration – pH change in the titration of a strong base with universal indicator 2. Study pH change by common-ion effect in weak acids and bases |
D. Chemical Equilibrium | Perform one of the following: – Study the equilibrium shift between ferric and thiocyanate ions with varying concentrations – Study equilibrium shift between [Co(H₂O)₆]²⁺ and chloride ions by altering ion concentrations |
E. Quantitative Estimation | 1. Use of mechanical/electronic balance 2. Preparation of a standard solution of Oxalic acid 3. Titration: NaOH solution vs. standard Oxalic acid 4. Preparation of standard Sodium Carbonate solution 5. Titration: HCl solution vs. standard Sodium Carbonate |
F. Qualitative Analysis | 1. Identification of one cation and one anion in a salt sample: Cations: Pb²⁺, Cu²⁺, Al³⁺, Fe³⁺, Mn²⁺, Ni²⁺, Zn²⁺, Co²⁺, Ca²⁺, Sr²⁺, Ba²⁺, Mg²⁺, NH₄⁺ Anions: CO₃²⁻, S²⁻, SO₃²⁻, NO₃⁻, NO₂⁻, Cl⁻, Br⁻, I⁻, SO₄²⁻, PO₄³⁻, CH₃COO⁻ (Insoluble salts excluded) 2. Detection of Nitrogen, Sulphur, and Chlorine in organic compounds |
Evaluation Scheme for CBSE 11th Chemistry Syllabus 2025-26
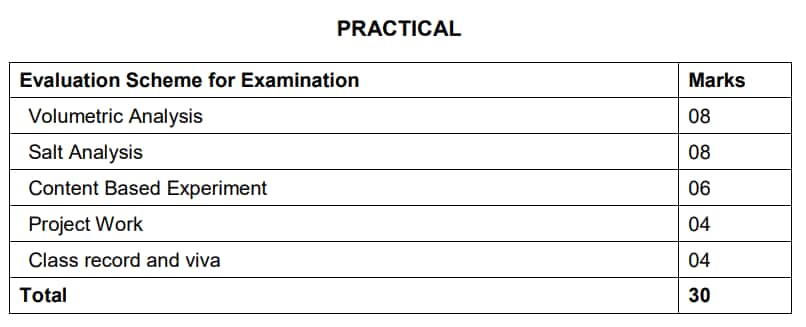
Focus on high-weightage units like Organic Chemistry and Thermodynamics.
Understand concepts thoroughly instead of rote learning. Concept clarity is required for atomic structure, bonding, and chemical equations.
Follow the NCERT book religiously. Cover all topics and stay consistent while studying.
Practice drawing neat diagrams for structures and apparatus.
Keep your practical file well-maintained throughout the academic year.
Revise topics weekly and focus on retaining information in a relaxed manner. Do not stress upon rote learning.
Also, check: CBSE Class 11 Syllabus 2025-26 (All Subjects)
Frequently Asked Questions (FAQs)
The latest CBSE Class 12 Physics syllabus and marking scheme 2025-26 is available at cbseacademic.nic.in. The download link to the syllabus PDF is also provided in the article above.
Optics (18 marks) and Magnetic Effects of Current and Magnetism (17 marks) carry the highest weightage.
On Question asked by student community
Hello Hashi,
Download the CBSE Class 12 Business Studies 2026 paper from the link below to prepare effectively.
https://school.careers360.com/boards/cbse/cbse-class-12-business-studies-question-paper-2026
Hello Gilla
You can download the question paper from the link given below:
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers-class-10-maths
Hope it helps.
Hello Student,
Check the article given below to access and download the CBSE question paper for classes 10 and 12.
Link:
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers
Dear Student,
You can appear for improvement exams in other passed subjects after clearning compartment exams. The Central Board of Secondary Education (CBSE) conducts the CBSE Class 12 improvement exams for students who are not satisfied with their scores in the Class 10 and Class 12 board examinations.
You can
Hi Madhuri,
For CBSE Class 12 Maths preparation, especially if your basics are weak, please refer to the link given below:
https://school.careers360.com/boards/cbse/cbse-class-12-maths-preparation-tips
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Apply for UG & PG programmes from Victoria University, Delhi NCR Campus
Admissions open for UG & PG programs at Illinois Tech Mumbai
Apply for UG & PG courses at University of Aberdeen, Mumbai Campus
UG & PG Admissions open for CS/AI/Business/Economics & other programmes.
Bristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes