Cells are the basic structural and functional units of life. They form tissues, organs, and systems, and perform essential processes like respiration, nutrition, and reproduction.
NCERT Solutions for Class 9 Science Chapter 2 - Cell: The Building Block of Life
NCERT Solutions for Class 9 Science Chapter 2 Cell The Building Block of Life answers many questions related to the cell. Why is a cell called the building block of life? How do cells divide and form new cells, or why do plant cells have a rigid cell wall, and animal cells do not? The answers are explained in these NCERT Solutions Class 9 Science Chapter 2. Cells are found everywhere, from the smallest bacterium to a complex human being. The Chapter 2 Exploration NCERT Solution Class 9 PDF direct download link is provided in this article to help students understand the key topics.
This Story also Contains
- NCERT Solutions for Class 9 Science Chapter 2 Cell: The Building Block of Life - Download PDF
- Class 9 Science Chapter 2 - Revise, Reflect, Refine Exercise Questions With Solutions
- Class 9 Science Chapter 2 Cell (In-text Questions): Think It Over, What if and Pause and Ponder
- What are the Important Topics Covered in Class 9 Science Chapter 2?
- How Do NCERT Solutions Help in Understanding Chapter 2 Cell?
- What If You Skip NCERT Solutions for Class 9 Science Chapter 2 Cell?

It includes NCERT solutions of class 9 science chapter 2 Exercise, which includes important questions, like how water movement affects seeds and potato pieces, prokaryotic vs eukaryotic cells, how mitosis helps in growth and meiosis for gamete formation in a simple way. This helps students prepare effectively for school exams and form the basics of biology for competitive exams. In accordance with CBSE guidelines, students can download the Exploration NCERT Chapter 2 Cell questions and answers PDF offline.
NCERT Solutions for Class 9 Science Chapter 2 Cell: The Building Block of Life - Download PDF
Students can download the complete updated NCERT questions with detailed answers for better learning. Cell: The Building Block of Life Class 9 questions and answers PDF include various topics that form the base of biology. The NCERT Solutions for Class 9 improve performance and boost confidence for exams.
Class 9 Science Chapter 2 - Revise, Reflect, Refine Exercise Questions With Solutions
All the answers and detailed solutions to the NCERT Revise, Reflect, Refine Exercise Questions given at the end of the chapter are given below. The Cell: The Building Block of Life class 9 question-answers form the base of many topics in science for higher classes.
Revise, Reflect, Refine
Question 1: Differentiate between the following pairs of terms based on the clues given in parentheses:
(i) Cell membrane and cell wall (permeability)
(ii) RER and SER (structure)
(iii) Chloroplasts and chromoplasts (pigments)
Answer: (i) Cell membrane vs Cell wall (permeability)
|
Cell membrane |
Cell wall |
|
It is selectively permeable, which allows only certain substances to pass |
It is fully permeable to most substances, which can pass freely. |
|
Thin, flexible covering around the cell. |
Thick, rigid covering outside the cell membrane. |
|
Present in both plant and animal cells |
Present only in plant cells. |
(ii) RER vs SER (structure)
|
RER |
SER |
|
It has ribosomes attached and appears rough. |
No ribosomes and appears smooth. |
|
Helps in protein synthesis |
Helps in lipid and fat synthesis. |
|
Found near the nucleus |
Found away from the nucleus. |
(iii) Chloroplasts vs Chromoplasts (pigments)
|
Chloroplasts |
Chromoplasts |
|
It contains only green chlorophyll pigments. |
It contains carotenoid pigments (yellow, orange, red). |
|
These pigments help in photosynthesis. |
These pigments provide colour to fruits and flowers. |
|
Found mainly in leaves |
Found mainly in petals and ripened fruits |
Question 2: Two similar animal cells are placed in two different solutions:
-
Cell X is placed in pure water.
-
Cell Y is placed in a concentrated salt solution.
Cells are observed after some time. Cell X swells, and Cell Y shrinks.
Which statement provides the correct explanation for the above observations?
(i) Salt molecules moved into Cell Y, causing it to shrink.
(ii) Water moved into Cell X, and more water moved out of Cell Y than the salt solution entered in it.
(iii) Water moved into Cell X and moved out of Cell Y through the cell membrane.
(iv) Solute movement caused osmosis in both cells.
Answer: The correct statement is: (iii) Water moved into Cell X and moved out of Cell Y through the cell membrane.
Explanation:
-
Water enters Cell X, placed in pure water by osmosis, making it swell.
-
In a concentrated salt solution, water leaves Cell Y by osmosis, causing it to shrink.
-
This happens because the cell membrane is selectively permeable, which allows water molecules to move but not salt molecules.
-
Therefore, the difference in water movement explains the swelling of Cell X and the shrinking of Cell Y.
Question 3: Look at the diagram of a cell in Fig. Identify the parts labelled from (a) to (g) and correctly match them with their functions given below:
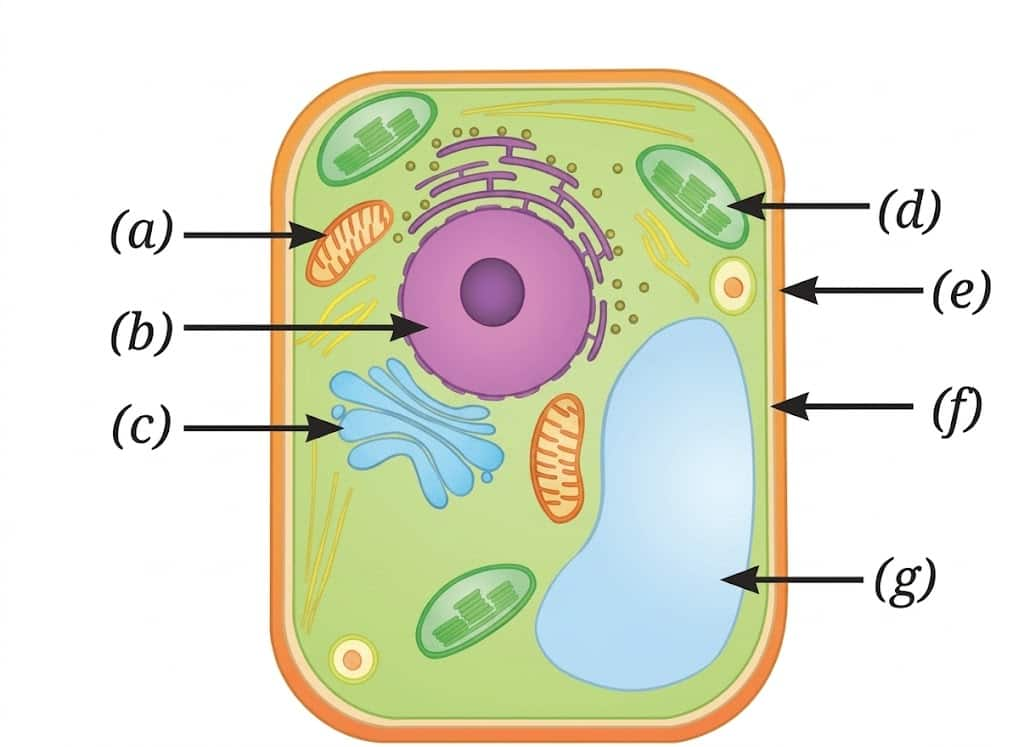
(i) Controlling all the activities of a cell.
(ii) Site of cellular respiration.
(iii) A storage organelle that also provides rigidity to the cell.
(iv) Separates the cell contents from the surroundings.
(v) Provides structural rigidity to the cell.
(vi) Packs and stores materials received from ER.
(vii) Helps in manufacturing food.
Answer:
|
Function |
Label |
Organelle |
|
(i) Controlling all the activities of a cell |
(b) |
Nucleus |
|
(ii) Site of cellular respiration |
(a) |
Mitochondria |
|
(iii) A storage organelle that also provides rigidity to the cell |
(g) |
Vacuole |
|
(iv) Separates the cell contents from the surroundings |
(f) |
Cell membrane |
|
(v) Provides structural rigidity to the cell |
(e) |
Cell wall |
|
(vi) Packs and stores materials received from ER |
(c) |
Golgi apparatus |
|
(vii) Helps in manufacturing food |
(d) |
Chloroplast |
Question 4: Which of the following option(s) of the pairs of cell organelles are correctly placed under the given categories?
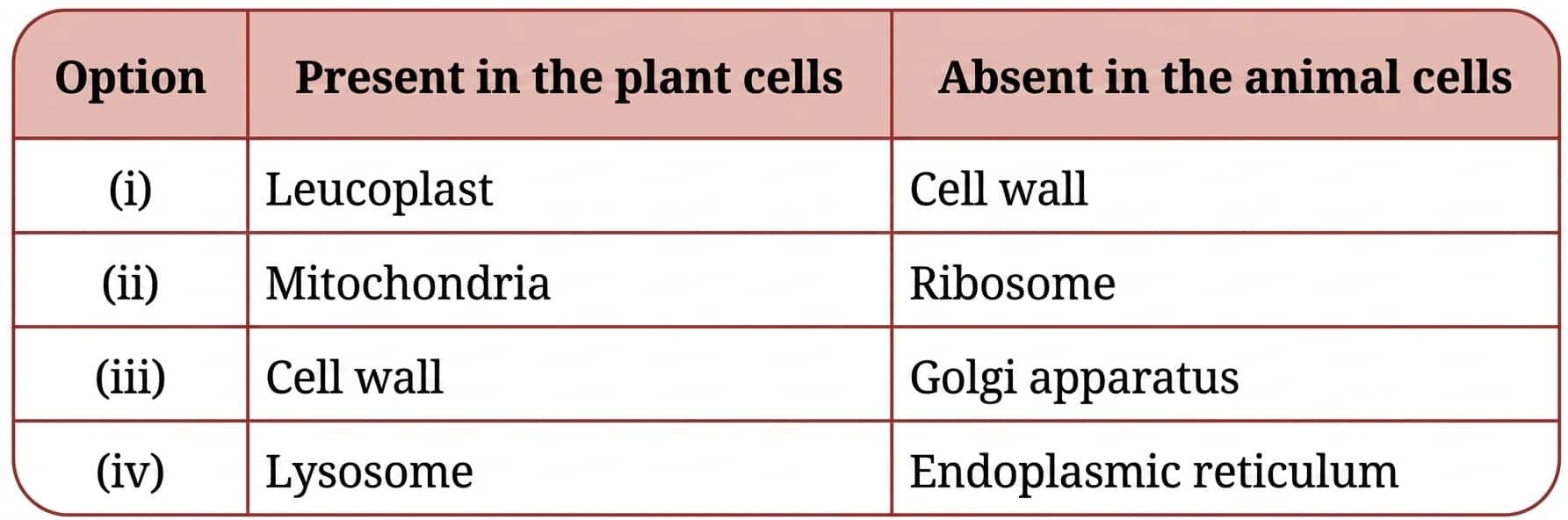
Answer:
|
Option |
Present in Plant Cells |
Absent in Animal Cells |
Correct? |
|
(i) |
Leucoplast |
Cell wall |
Yes, Correct |
|
(ii) |
Mitochondria |
Ribosome |
Wrong, as ribosomes are present in animal cells too |
|
(iii) |
Cell wall |
Golgi apparatus |
Wrong as the Golgi apparatus is present in animal cells |
|
(iv) |
Lysosome |
Endoplasmic reticulum |
Wrong as ER is present is animal cell also. |
Question 5: Two students, Renu and Rohit, were having a discussion on the plastids. Renu emphasised that all parts of the plants, even roots, contain plastids. However, Rohit did not agree with the statement and told her that plastids are absent in plant roots since the roots are underground and do not need to perform photosynthesis. Who is correct? Justify your answer.
Answer: Renu is more correct. Reason -
Plastids are of three types:
-
Chloroplasts - green, for photosynthesis.
-
Chromoplasts - colored, giving red/yellow/orange to flowers and fruits.
-
Leucoplasts - colourless, store starch, oils, and proteins.
-
Roots do not contain chloroplasts as they are underground and cannot photosynthesise. In this sense, Rohit is partly right.
-
But roots do contain leucoplasts, which are plastids specialised for storage. For example, potato tubers store starch in leucoplasts.
-
Therefore, plastids are present in roots, but in the form of leucoplasts, not chloroplasts.
Renu is correct overall because plastids exist in all plant parts and differ in their type. Rohit is only partly correct, as roots lack chloroplasts but still have leucoplasts.
Question 6: Mitochondria and chloroplasts are two important organelles in a plant cell. Discuss how these two organelles are structurally and functionally similar to each other, and different from each other.
Answer: Mitochondria and chloroplasts are structurally similar because both are double‑membrane organelles. They have their own DNA and ribosomes, but they differ in function. Mitochondria generate ATP through respiration, and chloroplasts produce glucose through photosynthesis.
Structural Similarities
-
Double membrane: Both have an outer and inner membrane.
-
Own DNA & ribosomes: They can synthesise some of their own proteins independently.
-
Internal compartments: Mitochondria have a matrix with enzymes. Chloroplasts have a stroma with enzymes.
-
Membrane folds: Mitochondria have cristae (increase surface area for respiration). Chloroplasts have thylakoid membranes (stacked into grana for photosynthesis).
Functional Similarities
-
Energy conversion organelles: Both are involved in energy modification.
-
ATP production: Both generate ATP (mitochondria via respiration, chloroplasts via photophosphorylation).
-
Semi‑autonomous: Both can divide and replicate within the cell.
Differences
|
Feature |
Mitochondria |
Chloroplasts |
|
Main function |
Break down glucose to release energy (ATP). |
Use sunlight to make glucose (photosynthesis). |
|
Pigments |
No pigments. |
Contain chlorophyll and other pigments. |
|
Location |
Found in both plant and animal cells. |
Found only in plant cells and algae. |
|
Membrane folds |
Cristae (inner folds). |
Thylakoids are stacked into grana. |
|
Energy source |
Organic molecules (glucose, fatty acids). |
Sunlight. |
Question 7: Which of the following pairs of cell organelles contains DNA?
(i) Chloroplasts, Ribosomes
(ii) Mitochondria, Nucleus
(iii) Golgi bodies, Ribosomes
(iv) Nucleus, Lysosomes
Answer: The correct option is: (ii) Mitochondria and Nucleus.
-
The nucleus contains the cell’s main DNA, which controls all activities.
-
Mitochondria have their own circular DNA and ribosomes, enabling them to produce some proteins independently.
-
Chloroplasts also contain DNA, but ribosomes do not.
-
Golgi bodies and lysosomes do not contain DNA.
Therefore, the pair that correctly contains DNA is the Mitochondria and Nucleus (option ii).
Question 8: A researcher carried out an experiment in which she took two carrots of similar size. She placed one carrot in plain water and the other carrot in a concentrated salt solution (Fig. ). After 24 hours, she recorded her observations.

Experimental set-up having a carrot (a) in plain water, and (b) in salt solution
(i) What hypothesis does she want to test through this experiment?
(ii) What would you suggest for the improvement of this experiment?
(iii) Why does the carrot in plain water stay stiff and crunchy, but the carrot in concentrated salt solution become rubbery and limp?
Answer: (i) Hypothesis:
The researcher wants to test whether osmosis (the movement of water across a selectively permeable membrane) affects plant tissues differently in pure water and concentrated salt solutions.
(ii) Improvement suggestion:
-
Use equal-sized carrot pieces instead of whole carrots for uniformity.
-
Keep them in equal volumes of solution under the same conditions.
-
Record changes in weight or length for more accurate results.
(iii) Explanation of observations:
-
In plain water, water enters carrot cells by osmosis, making the carrot turgid, and the carrot stays stiff and crunchy.
-
In a concentrated salt solution, water leaves carrot cells by osmosis, making them flaccid, and the carrot becomes rubbery and limp.
Question 9: Indicate the presence or absence of the following structures in bacterial and animal cells:
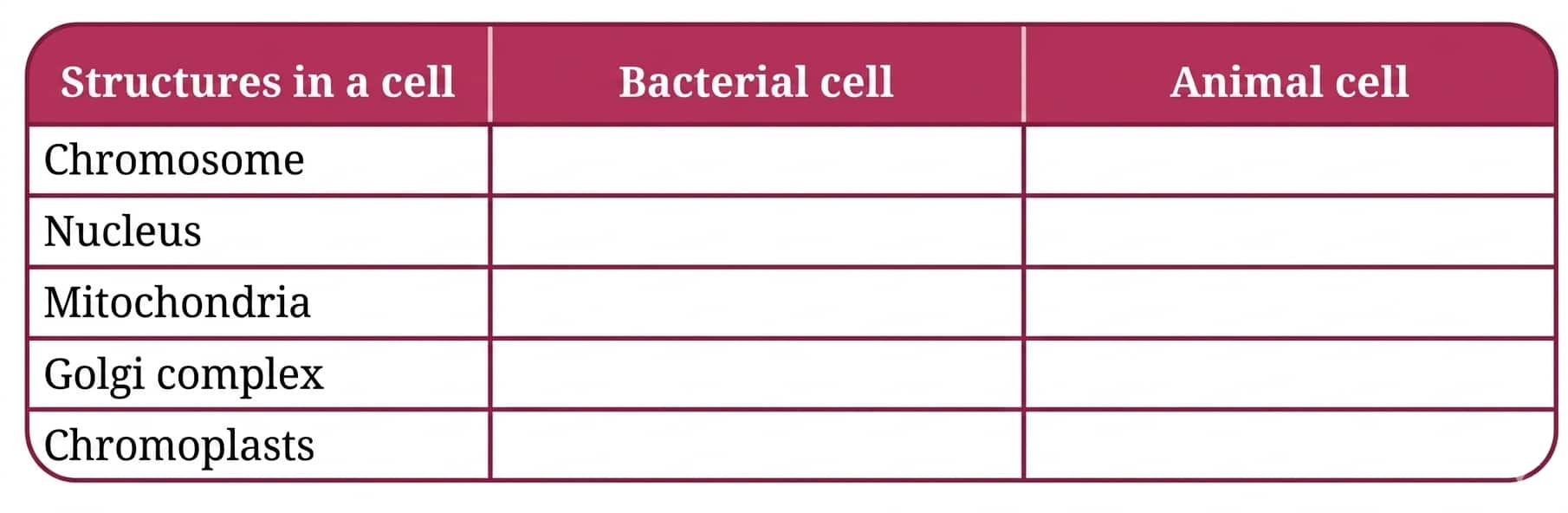
Answer:
|
Structure |
Bacterial Cell |
Animal Cell |
|
Chromosome |
Present (single, circular DNA) |
Present (multiple, linear DNA) |
|
Nucleus |
Absent (DNA lies in the nucleoid region) |
Present (true nucleus with nuclear membrane) |
|
Mitochondria |
Absent |
Present |
|
Golgi complex |
Absent |
Present |
|
Chromoplasts |
Absent |
Absent (chromoplasts are only in plant cells) |
Question 10: Carry out the following experiment:
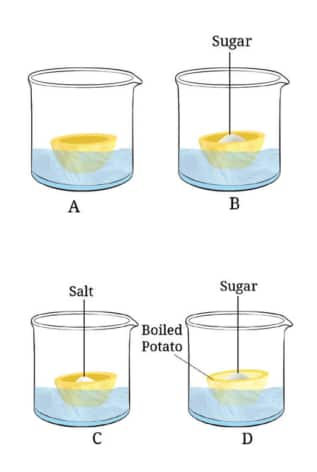
Take four peeled potato halves and scoop each one out to make potato cups. One of these potato cups should be made from a boiled potato. Place each of the potato cups in a beaker containing water (Fig. ). Now, set up the experiment as follows:
(a) Keep Cup A empty.
(b) Add one teaspoon sugar in Cup B.
(c) Add one teaspoon salt in Cup C.
(d) Add one teaspoon sugar in the boiled potato in Cup D.
Observe the four potato cups at least two hours and answer the following questions:
(i) Explain why water gathers in the hollowed portion of Cup B and Cup C.
(ii) Why is Cup A necessary for this experiment?
(iii) Explain why water does not gather in the hollowed portions of Cups A and D.
Answer: (i) Why water gathers in Cup B and Cup C:
-
Sugar in Cup B and salt in Cup C create a concentrated solution inside the potato cup.
-
Water from the beaker moves into the hollowed portion by osmosis through the potato tissue.
(ii) Why Cup A is necessary:
-
Cup A (empty) acts as a control experiment.
-
It shows that without sugar or salt, no water collects in the hollow, proving that osmosis happens only when a concentration difference exists.
(iii) Why water does not gather in Cup A and Cup D:
-
Cup A have no solute inside, so there is no concentration difference. Hence, no osmosis.
-
Cup D is made from boiled potatoes. So its cells are dead, and membranes are destroyed. Hence, osmosis cannot occur.
Question 11: Identify the pair that incorrectly matches the cell organelle with its function.
(i) Ribosome — Protein synthesis
(ii) SER — Lipid and cellulose synthesis
(iii) Lysosome — Digestion of foreign agents
Answer:
The incorrect pair is: (ii) SER — Lipid and cellulose synthesis.
(i) Ribosome - Protein synthesis: Correct, they synthesise proteins.
(ii) SER - Lipid and cellulose synthesis: Incorrect as SER makes lipids and detoxifies chemicals, but does not synthesise cellulose. Cellulose is made by enzymes at the cell wall, not by SER.
(iii) Lysosome - Digestion of foreign agents: Correct, they digest foreign agents and worn‑out cell parts.
Therefore, the mismatch is SER - Lipid and cellulose synthesis.
Question 12: What outcome do you expect if all the mitochondria are removed from a eukaryotic cell?
Answer: If all the mitochondria are removed from a eukaryotic cell:
-
The cell will not be able to produce energy (ATP) through respiration.
-
Without ATP, the cell cannot carry out essential activities like growth, division, transport, or repair.
-
The cell will eventually die because mitochondria are the “powerhouses” of the cell.
Without mitochondria, the cell cannot make energy and will stop functioning.
Question 13: Which phenomenon inhibits the formation of tumours in the human body? Can plants also develop tumours? Explain
Answer: Phenomenon that inhibits tumour formation:
-
The phenomenon is contact inhibition.
-
Normal cells stop dividing when they touch neighbouring cells. This prevents uncontrolled growth and protects the body from tumour formation.
If the human body develops loss of contact inhibition, the cells keep dividing even after touching each other. It forms an uncontrolled mass of cells called a tumour. In cancer cells, this property is lost.
Can plants develop tumours?
-
Yes, plants can also develop tumours.
-
Example: Crown gall disease caused by Agrobacterium tumefaciens.
-
Plant tumours are usually localised and do not spread to other parts like malignant tumours in animals.
-
They often result from infections, injury, or abnormal cell division.
Question 14: The cell membrane of a cell is made up of proteins and lipids. Which cell organelles help in the synthesis of cell membrane? Write the path of these compounds from their site of synthesis to the cell membrane and show this through a labelled diagram.
Answer: The cell membrane is made of proteins and lipids. Cell organelles involved in cell membrane synthesis:
-
Smooth Endoplasmic Reticulum (SER) synthesises lipids.
-
Rough Endoplasmic Reticulum (RER) synthesises proteins with the help of ribosomes.
-
The Golgi apparatus modifies, packs, and transports proteins and lipids.
This provides that the cell membrane is continuously formed and repaired.
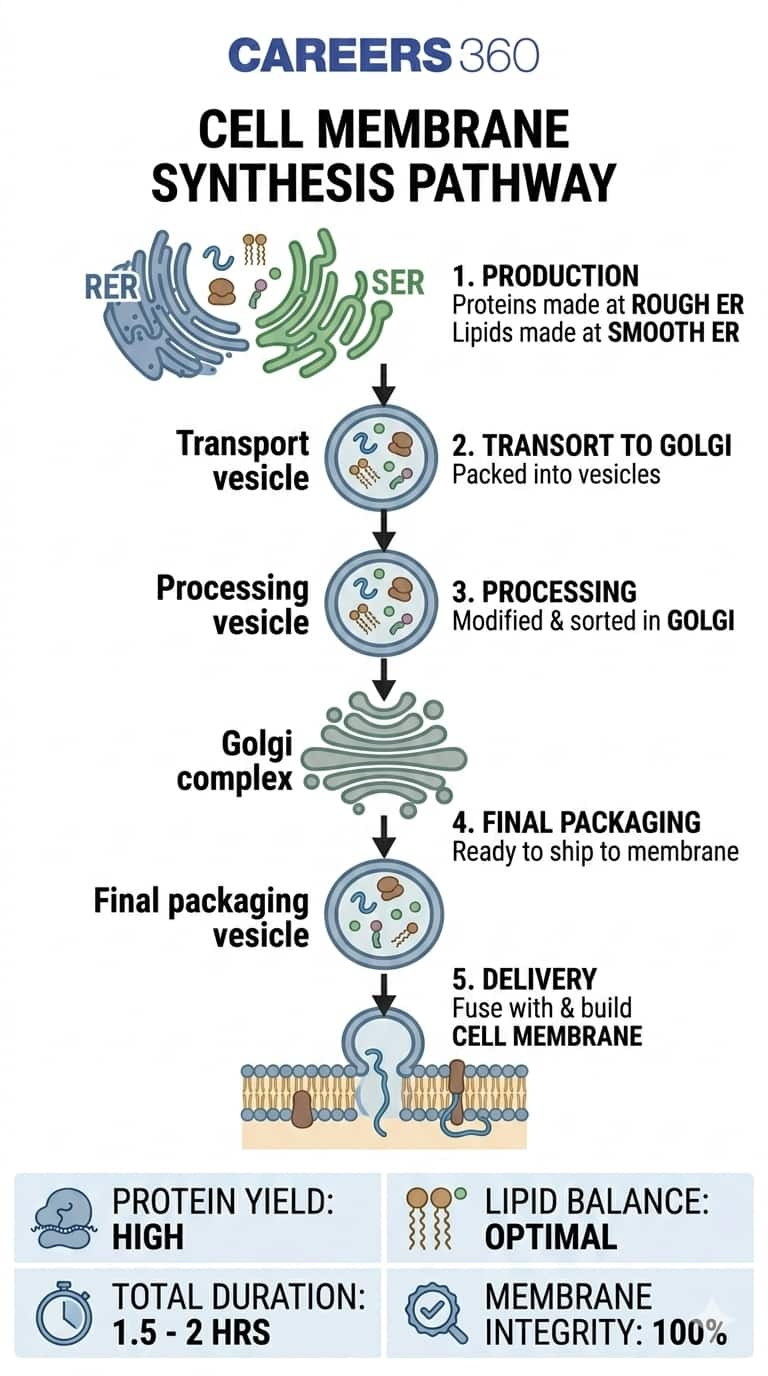
Pathway of Synthesis to the Cell Membrane:
-
Proteins are made in the RER (with ribosomes).
-
Lipids are made in SER.
-
Both are sent to the Golgi apparatus for processing and packaging.
-
Golgi vesicles carry them to the cell membrane, where they are assembled into the membrane structure.
Question 15: What would happen if gametes are formed by mitotic divisions?
Answer: If gametes were formed by mitotic divisions instead of meiosis:
-
Gametes would have the same number of chromosomes as the parent cell (diploid, 2n).
-
When two such gametes fuse during fertilisation, the resulting zygote would have double the chromosome number (4n).
-
This would disturb the chromosome number balance in every generation.
-
As a result, the species would not maintain a stable chromosome number, and normal reproduction would be affected.
Gametes must be formed by meiosis to keep the chromosome number constant across generations. If formed by mitosis, the chromosome number would keep doubling, and life could not continue normally.
Question 16: A farmer, Deepa, was very happy with the harvest of amla (Indian Gooseberry) and lemons on her farm. However, she could sell only one-fourth of the produce in the local market. Recognising that a significant amount of produce may be lost post-harvest, she employed a traditional yet scientifically sound method to extend the shelf life of amla and lemons. She turned perishable produce into profitable products, such as pickles and sharbat. She used the excess produce to prepare pickles, murabbas, and sharbat by adding appropriate amounts of salt, sugar, or jaggery to small pieces of fruit and their juices. These were then stored in small glass bottles for sale, helping her prevent the wastage of post-harvest produce. This shift from farming to agro-processing would strengthen food security and boost the local economy, creating a sustainable model that cuts waste while increasing her income. Based on the above passage answer the following questions:
(i) Which scientific concept has the farmer applied in the preservation of the farm produce?
(ii) How does the addition of high concentrations of salt and sugar create an environment that prevents the growth of spoilage-causing bacteria and fungi?
(iii) Suggest a healthy recipe of this kind for food preservation.
(iv) What are the scientific values addressed in this case?
Answer: (i) Scientific concept applied:
-
The farmer used food preservation by adding high concentrations of salt, sugar, or jaggery.
-
This is based on the principle of osmosis, which prevents microbial growth.
(ii) Role of salt and sugar:
-
High concentrations of salt or sugar create a hypertonic environment.
-
Water moves out of microbial cells, and microbes lose water and cannot survive.
-
This prevents the growth of bacteria and fungi that cause spoilage.
(iii) Healthy recipe suggestion:
-
Amla candy: Cut amla into pieces, boil lightly, soak in sugar syrup, dry in sunlight, and store. This preserves nutrients and extends shelf life.
-
Other examples: lemon pickle with salt, jaggery‑based amla murabba.
(iv) Scientific values addressed:
-
Food security prevents wastage of farm produce.
-
Sustainability converts perishable items into long‑lasting products.
-
Economic growth increases farmers’ income through agro‑processing.
-
Scientific awareness applies traditional methods with scientific reasoning.
Confused between CGPA and Percentage?
Get your results instantly with our calculator!
Class 9 Science Chapter 2 Cell (In-text Questions): Think It Over, What if and Pause and Ponder
The detailed solution to all these in-text exercises in this chapter is given below. Studying the NCERT Solutions Class 9 Science Chapter 2 Cell Think It Over, What If, and Pause & Ponder questions helps students to explore how cells were discovered, and why they are called the building blocks of cells.
Think It Over (Page 8)
Question 1: Where does a cell come from?
Answer: Cells originate from pre-existing cells through the process of cell division (cell theory). It was given by Rudolf Virchow in 1855, who stated it as “Omnis cellula e cellula”, meaning every cell comes from a cell.
Question 2: How have technological interventions facilitated the creation of new knowledge in understanding the world beyond the naked eye?
Answer: Technological tools like microscopes have helped scientists see things too small for the human eye. The first observation of cells was done using a light microscope. The electron microscope showed detailed structures like mitochondria and ribosomes. Staining techniques also made cell parts clearer. These interventions gave us new knowledge about the cell structure, its organelles, and how it functions, which was not possible with the naked eye.
Question 3: How is the cell the structural and functional unit of life?
Answer: A cell is called the structural and functional unit of life because:
-
All living organisms are made up of cells.
-
Cells provide structure by forming tissues, organs, and organ systems.
-
Cells perform basic functions such as respiration, nutrition, excretion, and reproduction.
-
In multicellular organisms, every activity of the body begins at the cellular level.
Thus, the cell is the smallest unit capable of carrying out all life processes, making it both the structural and functional unit of life.
Question 4: How does a cell multiply?
Answer: A cell multiplies through the process of cell division.
-
In mitosis, one cell divides into two identical daughter cells. This helps in the growth, repair, and replacement of old cells.
-
In meiosis, a cell divides to form four daughter cells with half the chromosome number. This is important for sexual reproduction and the formation of gametes.
Thus, cell multiplication ensures continuity of life, growth of organisms, and transfer of genetic information to the next generation.
What If (Page 12)
Question 1: What if mung bean seeds are kept in a concentrated solution after soaking in water for 12 hours?
Answer: When mung bean seeds are placed in a concentrated solution after soaking, they lose water by osmosis. The water inside the seed cells moves out into the concentrated solution, causing the cells to shrink. As a result, the seeds become shrivelled, and their growth or germination slows down. This shows how the movement of water across a cell membrane affects living cells.
Pause and Ponder
Page 14
Question 1: What argument would you give for the necessity of a cell wall in plants usually fixed in one place versus in animals usually moving from one place to the other?
Answer: Plants are fixed in one place and cannot move to avoid stress, so they need a cell wall for strength and protection. The rigid wall helps them stand upright, resist wind, and maintain shape even under water pressure. Animals move freely and have flexible bodies, so they do not need a rigid cell wall.
Question 2: What consequences would you predict for a plant cell if its cell wall were to become as flexible as a cell membrane?
Answer: If the cell wall became as flexible as the cell membrane, the plant cell would lose rigidity. It could swell or burst when water enters, and the plant would not be able to stand upright. This would make plants weak, floppy, and unable to survive strong environmental conditions.
Question 3: Why is it important to cut the two potato pieces in roughly equal size and measure their initial weight before placing them in different liquids?
Answer: Equal size and initial weight ensure a fair comparison in the osmosis experiment. If the potato pieces are unequal, changes in weight or size may be due to their difference rather than the effect of the liquids. Measuring before and after shows the real impact of osmosis.
Page 19
Question 4: Do white flowers contain any pigment? Give reasons.
Answer: Yes, white flowers do contain pigments. They have flavonoids (like flavones and flavonols), which are colourless or pale. These pigments scatter light and give the flower a white appearance. So, even though they look white, they are not completely pigment‑free.
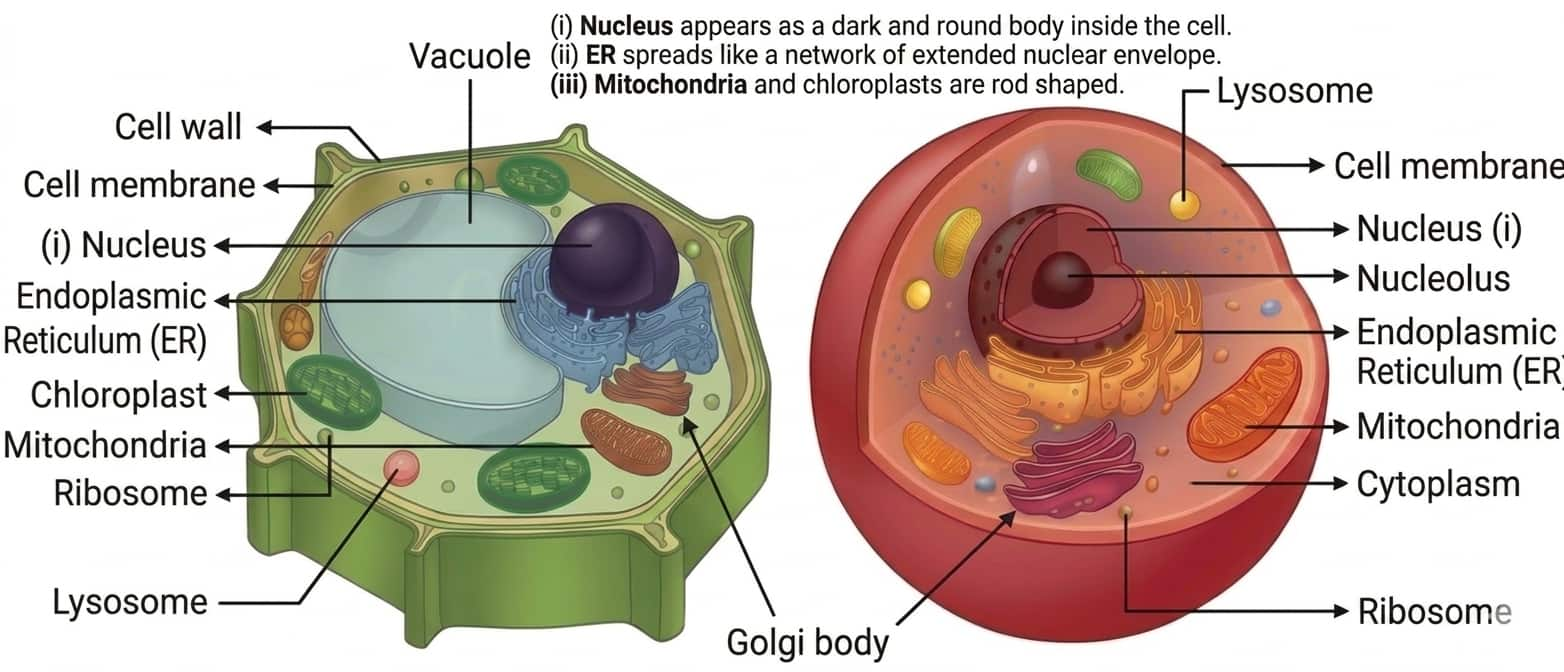
Question 5: Draw a well-labelled schematic diagram of a plant or an animal cell using these clues —
(i) Nucleus appears as a dark and round body inside the cell.
(ii) ER spreads like a network of extended nuclear envelope.
(iii) Mitochondria and chloroplasts are rod shaped.
Answer:
Page 22
Question 6: Instead of many small ones, why does a cell not have a single giant mitochondrion? How does this relate to the concept of surface area?
Answer: A cell has many mitochondria instead of one giant mitochondrion because small mitochondria provide a larger total surface area. The inner membrane of mitochondria has folds (cristae) where energy production occurs. More mitochondria mean more surface area for respiration, which increases efficiency. A single giant mitochondrion would have less surface area compared to many small ones, reducing energy output.
Question 7: If the skin cells start dividing by meiosis instead of mitosis, what do you think will happen to a cut on the skin?
Answer: If skin cells divide by meiosis, they will produce cells with half the chromosome number. These cells cannot replace normal skin cells. As a result, the cut will not heal properly, and the skin tissue will fail to repair itself. Healing requires identical cells formed by mitosis, not gametes formed by meiosis.
What are the Important Topics Covered in Class 9 Science Chapter 2?
Important topics and subtopics of this chapter are listed below. Questions are frequently asked about these topics in the exam. To gain clarity, students can refer to the NCERT Solutions for Class 9 Science Chapter 2 Cell.
-
Plasma Membrane or Cell Membrane
-
Mitochondria
-
Plastids
-
Endoplasmic Reticulum (RER and SER)
-
Differences Between Plant and Animal Cells
-
Processes: Diffusion, Osmosis, Plasmolysis
-
Cell Division: Mitosis and Meiosis
How Do NCERT Solutions Help in Understanding Chapter 2 Cell?
Cells are the basic structural and functional units of all living organisms. This chapter explains their importance and how they work together to support life.
-
Students will understand the concept of the cell and its discovery with the help of the Cell: The Building Block of Life Class 9 question answer.
-
They will learn about different cell organelles and their functions, along with the diagrams.
-
The solutions explain the differences between plant and animal cells in a simple way.
-
They will know how the processes of mitosis and meiosis take place, by which cells divide and grow.
-
With Class 9 Science Cell: The Building Block of Life question answers, learners can understand important diagrams like cell structure.
What If You Skip NCERT Solutions for Class 9 Science Chapter 2 Cell?
If you skip the NCERT Solutions for Class 9 Science Chapter 2 Cell, you may face these problems:
-
The cell is the foundation of biology. So without NCERT practice, you may not understand the basics of organelles, cell theory, or the differences between plant and animal cells.
-
NCERT questions repeatedly come directly in school exams, so skipping solutions means you miss the exact wording and style of answers expected.
-
NCERT solutions explain answers step by step. Without them, you may feel confused during revision, leading to weak confidence
-
This Class 9 Science Cell chapter builds the base for higher classes and competitive exams like NEET. So, missing it now makes later topics harder.
Frequently Asked Questions (FAQs)
Osmosis is the movement of water across a selectively permeable membrane. Class 9 NCERT experiments with potato cups, carrot pieces, and seeds showed how water movement affects cells.
Cells divide by mitosis (growth and repair) and meiosis (formation of gametes). This provides continuity of life and equal chromosome numbers across generations.
Popular Questions
Applications for Admissions are open.
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Victoria University, Delhi NCR
ApplyApply for UG & PG programmes from Victoria University, Delhi NCR Campus
Illinois Tech Mumbai
ApplyAdmissions open for UG & PG programs at Illinois Tech Mumbai
University of Aberdeen Mumbai
ApplyApply for UG & PG courses at University of Aberdeen, Mumbai Campus
University of York, Mumbai
ApplyUG & PG Admissions open for CS/AI/Business/Economics & other programmes.
University of Bristol, Mumbai Enterprise Campus
ApplyBristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes

