JEE Main Important Physics formulas
As per latest syllabus. Physics formulas, equations, & laws of class 11 & 12th chapters
The CBSE 11th chemistry exam is conducted for a total of 100 marks, out of which 70 marks are assigned to the theory paper and the remaining 30 marks are for the practical paper. The Central Board of Secondary Education (CBSE) has released the CBSE 11th Chemistry lab manual 2026 based on the NCERT curriculum. The CBSE 11th Chemistry lab manual 2026 is the official guide for students, as it outlines experiments, observations, procedures, safety measures, material required, etc. Students appearing for the exam can visit the NCERT official website, ncert.nic.in, to download the CBSE 11th Chemistry lab manual PDF 2026. Read further to learn in detail about the CBSE 11th Chemistry lab manual.
This Story also Contains
Also, check
Students appearing for the CBSE Class 11th examination 2026 must score passing marks in both theory and practical papers. Below, we have provided direct links to download the CBSE 11th Chemistry lab manual PDF 2026, for reference. They can use it to know the theory, objective and other information related to the practical so they can perform well in the CBSE practical exam 2026 for Class 11th Chemistry.
Sample Image of CBSE 11th Chemistry Lab Manual 2026


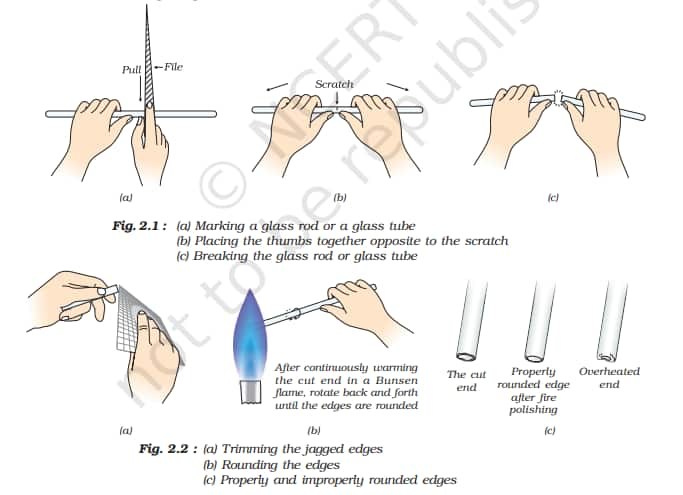

Micro-chemical methods are available for several of the practical experiments; wherever possible, such techniques should be used.
Basic Laboratory Techniques
1. Cutting glass tube and glass rod
2. Bending a glass tube
3. Drawing out a glass jet
4. Boring a cork
Characterisation and Purification of Chemical Substances
1. Determination of the melting point of an organic compound.
2. Determination of the boiling point of an organic compound.
3. Crystallisation of an impure sample of any one of the following: Alum, Copper Sulphate, Benzoic Acid.
Experiments based on pH
1. Any one of the following experiments:
Determination of pH of some solutions obtained from fruit juices, solutions of known and varied concentrations of acids, bases and salts using pH paper or universal indicator.
Comparing the pH of solutions of strong and weak acids of the same concentration.
Study the pH change in the titration of a strong base using a universal indicator.
2. Study the pH change by common-ion in case of weak acids and weak bases.
Chemical Equilibrium
Any one of the following experiments:
Study the shift in equilibrium between ferric ions and thiocyanate ions by increasing/decreasing the concentration of either of the ions.
Study the shift in equilibrium between [Co(H2O)6] 2+ and chloride ions by changing the concentration of either of the ions.
Quantitative Estimation
1. Using a mechanical balance/electronic balance.
2. Preparation of a standard solution of Oxalic acid.
3. Determination of the strength of a given solution of Sodium hydroxide by titrating it against a standard solution of Oxalic acid.
4. Preparation of a standard solution of Sodium carbonate.
5. Determination of the strength of a given solution of hydrochloric acid by titrating it against a standard Sodium Carbonate solution.
Qualitative Analysis
1. Determination of one anion and one cation in a given salt Cations: ???+,???+, ???+, ???+,???+, ???+, ???+, ???+, ???+, ???+, ???+, ???+, ??? + Anions: ??? ?−,? ?− , ??? ?−, ??? −, ??? − , ??−, ??−, ? − ,??? ?−, ??? ?− , ??????− (Note: Insoluble salts excluded)
2. Detection of -Nitrogen, Sulphur, Chlorine in organic compounds.
Also, read-
Get your results instantly with our calculator!
Scientific investigations involve laboratory testing and collecting information from other sources.
A few suggested Projects
a) Checking the bacterial contamination in drinking water by testing the sulphide ion
b) Study of the methods of purification of water
c) Testing the hardness, presence of Iron, Fluoride, Chloride, etc., depending upon the regional variation in drinking water and study of causes of presence of these ions above the permissible limit (if any).
d) Investigation of the foaming capacity of different washing soaps and the effect of the addition of Sodium carbonate on it
e) Study the acidity of different samples of tea leaves.
f) Determination of the rate of evaporation of different liquids
g) Study the effect of acids and bases on the tensile strength of fibres.
h) Study of the acidity of fruit and vegetable juices.
Note: Any other investigatory project, involving approximately 10 periods of work, can be chosen with the teacher's approval.
The CBSE board conducts the practical examination for 30 marks. Refer to the table below for the detailed marking scheme.
Evaluation scheme for examination | Marks |
Volumetric analysis | 08 |
Salt analysis | 08 |
Content-based experiment | 06 |
Project work | 04 |
Class record and viva | 04 |
Total | 30 |
Frequently Asked Questions (FAQs)
Students can find the CBSE 11th chemistry lab manual online in a PDF format at ncert.nic.in.
The CBSE 11th Chemistry practical exam will carry 30 marks in total.
On Question asked by student community
Hello Yuva,
Download CBSE Class 10 previous year papers from the link below to prepare effectively for all subjects.
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers-class-10
Hi Rudra,
Download the subject-wise CBSE 2nd exam paper from the link given in the article below.
Link:
https://school.careers360.com/boards/cbse/cbse-10th-second-exam-question-paper-2026
Hello,
Download the complete CBSE 10th maths question paper PDF of both maths basic and standard with complete solution by going through the article given below.
Link:
https://school.careers360.com/boards/cbse/cbse-class-10-maths-question-paper-2026
Hello,
Download the CBSE 10th question paper subject-wise from the link given below.
Link:
https://school.careers360.com/boards/cbse/cbse-class-10-question-paper-2026
https://school.careers360.com/boards/cbse/cbse-10th-second-exam-question-paper-2026
Hello Yash,
Download the Class 10 competency-based Questions from the article given below
Link:
https://school.careers360.com/boards/cbse/cbse-class-10-science-competency-based-questions-2026
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Apply for UG & PG programmes from Victoria University, Delhi NCR Campus
Admissions open for UG & PG programs at Illinois Tech Mumbai
Apply for UG & PG courses at University of Aberdeen, Mumbai Campus
UG & PG Admissions open for CS/AI/Business/Economics & other programmes.
Bristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes