University of Liverpool, Bengaluru Campus
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
The Central Board of Secondary Education has prescribed the CBSE Class 12 Chemistry practical 2026 project ideas and evaluation scheme. Students can visit the CBSE Board official website at cbseacademic.nic.in to download the project ideas for the CBSE practical exam for the 12th Class Chemistry. To study the presence of oxalate ions, to study the quantity of casein, to study the effect of potassium bisulphate, preparation of soybean milk, comparative study of the fermentation rate, etc., are some of the project ideas.
This Story also Contains

Surface chemistry, chemical kinetics, thermochemistry, electrochemistry, chromatography, preparation of inorganic compounds and organic compounds, qualitative analysis, etc., are some of the CBSE 12th syllabus for the Chemistry practical subject. The CBSE 12th board exam practicals were held from January 1 to 15, 2026. Read the article to learn more about the CBSE Class 12 Chemistry practical 2026, project ideas, and evaluation scheme.
Scientific investigations involve laboratory testing and collecting information from other sources. Check out a few suggested CBSE Board Class 12th Chemistry project ideas below.
Study of the presence of oxalate ions in guava fruit at different stages of ripening.
Study of the quantity of casein present in different samples of milk.
Preparation of soybean milk and its comparison with natural milk with respect to curd formation, effect of temperature, etc.
Study of the effect of Potassium Bisulphate as a food preservative under various conditions (temperature, concentration, time, etc.)
Study of the digestion of starch by salivary amylase and the effect of pH and temperature on it.
Comparative study of the rate of fermentation of the following materials: wheat flour, gram flour, potato juice, carrot juice, etc.
Extraction of essential oils present in Saunf (aniseed), Ajwain (carom), and Illaichi (cardamom).
Study of common food adulterants in fat, oil, butter, sugar, turmeric powder, chilli powder and pepper.
Students can check out the table below to learn more about the evaluation scheme for the CBSE Class 12 Chemistry practical exam.
Evaluation Scheme for Examination | Marks |
Volumetric Analysis | 8 |
Salt Analysis | 8 |
Content-Based Experiment | 6 |
Project Work | 4 |
Class record and viva | 4 |
Total | 30 |
Students are required to attend the viva-voce for the CBSE 12th Chemistry practical exam. External examiners ask the viva questions to students after the practical examination. Check out some of the practical viva sample questions for the CBSE Class 12 Chemistry subject.
Q1) What is titration?
Ans. The process of adding one solution from a burette to another in a conical flask to complete a chemical reaction is known as titration.
Q2) What is the basicity of H₂SO₄?
Ans. The basicity of H₂SO₄ is 2.
Q3) What are acidimetry and alkalimetry?
Ans. The methods of volumetric analysis involve the chemical reaction between an acid and a base to determine their concentrations are known as acidimetry and alkalimetry.
Q4) Is sodium hydroxide a primary standard?
Ans. No, sodium hydroxide is not a primary standard. It is a secondary standard.
Q5) What will be the normality of 0.10M KMnO₄?
Ans. The normality of KMnO₄ will be 0.1 x 5 = 0.5 N.
Get your results instantly with our calculator!
Students can check out the CBSE 12th Chemistry syllabus for the practical examination. Micro-chemical methods are available for several of the practical experiments; wherever possible, such techniques should be used.
Preparation of one lyophilic and one lyophobic sol
Lyophilic sol - starch, egg albumin and gum
Lyophobic sol – aluminium hydroxide, ferric hydroxide, arsenous sulphide.
Dialysis of sol-prepared in (a) above.
Study of the role of emulsifying agents in stabilising the emulsion of different oils.
Effect of concentration and temperature on the rate of reaction between Sodium Thiosulphate and Hydrochloric acid. Study of reaction rates of any one of the following:
Reaction of Iodide ion with Hydrogen Peroxide at room temperature using different concentrations of Iodide ions.
Reaction between Potassium Iodate (KIO3) and Sodium Sulphate: (Na2SO3) using starch solution as an indicator (clock reaction).
Any one of the following experiments
Enthalpy of dissolution of Copper Sulphate or Potassium Nitrate.
Enthalpy of neutralisation of strong acid (HCL) and strong base (NaOH).
Determination of the enthalpy change during interaction (Hydrogen bond formation) between Acetone and Chloroform.
![]()
Separation of pigments from extracts of leaves and flowers by paper chromatography and determination of Rf values.
Separation of constituents present in an inorganic mixture containing two cations only (constituents having a large difference in Rf values to be provided).
Preparation of double salt of Ferrous Ammonium Sulphate or Potash Alum.
Preparation of Potassium Ferric Oxalate.
Preparation of any one of the following compounds
Acetanilide
Di -benzalAcetone
p-Nitroacetanilide
Aniline yellow or 2-Naphthol Aniline dye.
Unsaturation, alcoholic, phenolic, aldehydic, ketonic, carboxylic and amino (Primary) groups.
Oxalic acid,
Ferrous Ammonium Sulphate
(Students will be required to prepare standard solutions by weighing themselves).
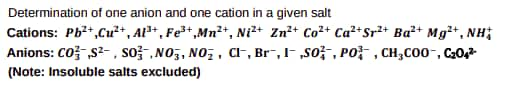
Frequently Asked Questions (FAQs)
The CBSE board has allotted 30 marks for the 12th Chemistry 2026 practical examination.
Students can visit the official website at cbseacademic.nic.in to check and download the CBSE Class 12 Chemistry project ideas from the syllabus file.
On Question asked by student community
Hi!
Given below is the link to access CBSE Class 12 Sociology Question Paper 2026 with Answer Key:
https://school.careers360.com/boards/cbse/cbse-class-12-sociology-answer-key-2026
Hello Hashi,
Download the CBSE Class 12 Business Studies 2026 paper from the link below to prepare effectively.
https://school.careers360.com/boards/cbse/cbse-class-12-business-studies-question-paper-2026
Hello Gilla
You can download the question paper from the link given below:
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers-class-10-maths
Hope it helps.
Hello Student,
Check the article given below to access and download the CBSE question paper for classes 10 and 12.
Link:
https://school.careers360.com/boards/cbse/cbse-previous-year-question-papers
Dear Student,
You can appear for improvement exams in other passed subjects after clearning compartment exams. The Central Board of Secondary Education (CBSE) conducts the CBSE Class 12 improvement exams for students who are not satisfied with their scores in the Class 10 and Class 12 board examinations.
You can
Study at a world-renowned UK university in India | Admissions open for UG & PG programs.
Apply for UG & PG programmes from Victoria University, Delhi NCR Campus
Admissions open for UG & PG programs at Illinois Tech Mumbai
Apply for UG & PG courses at University of Aberdeen, Mumbai Campus
UG & PG Admissions open for CS/AI/Business/Economics & other programmes.
Bristol's expertise meets Mumbai's innovation. Admissions open for UG & PG programmes